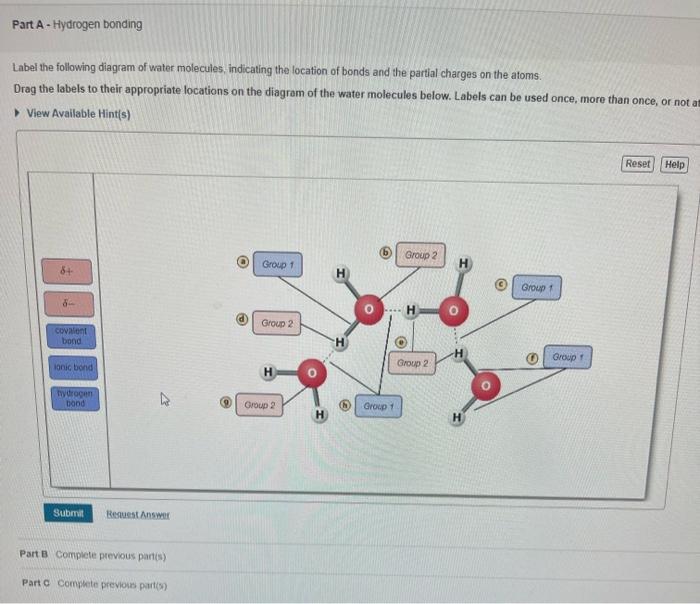
Draw A Water Molecule And Label The Partial Charges
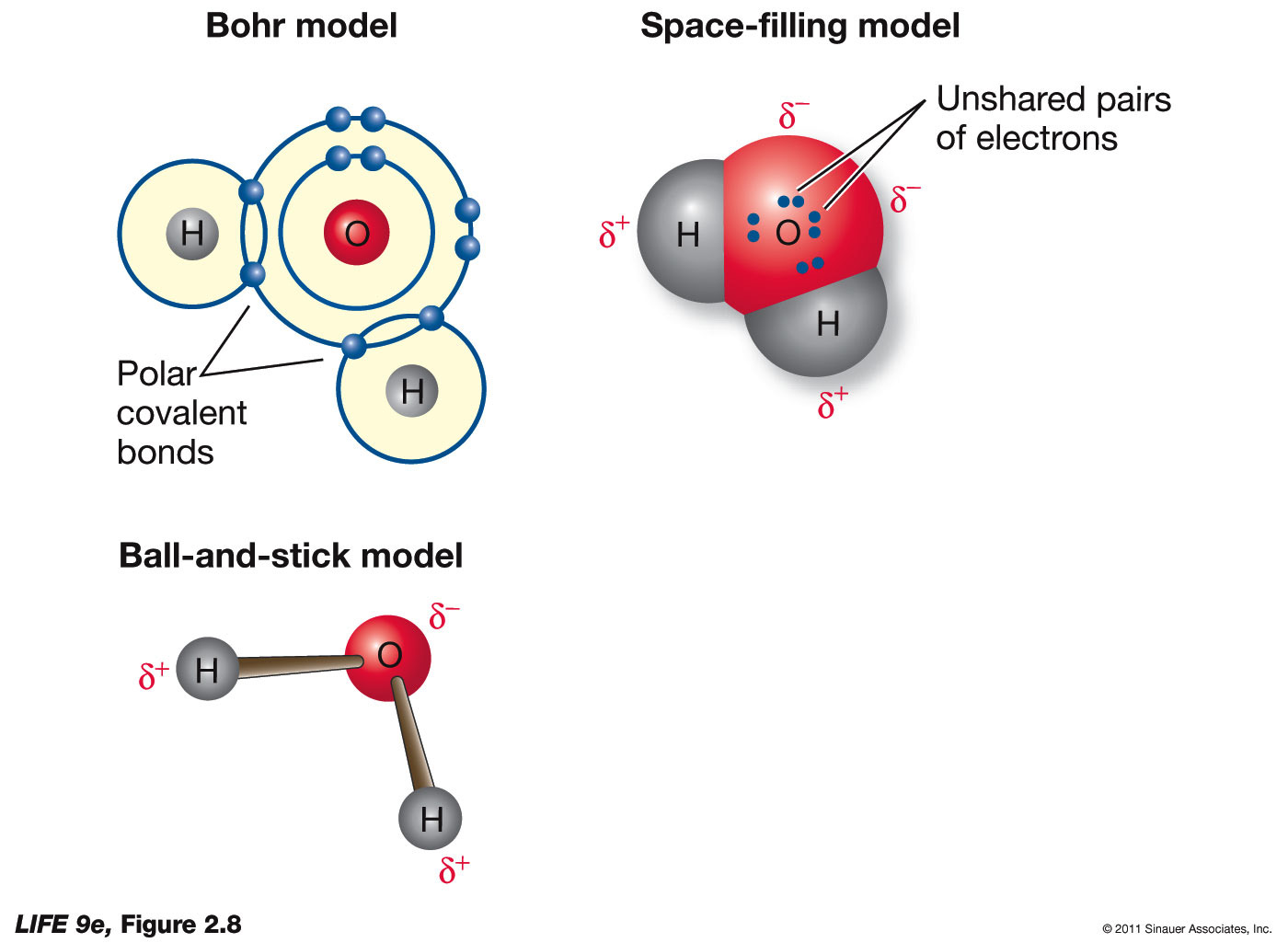
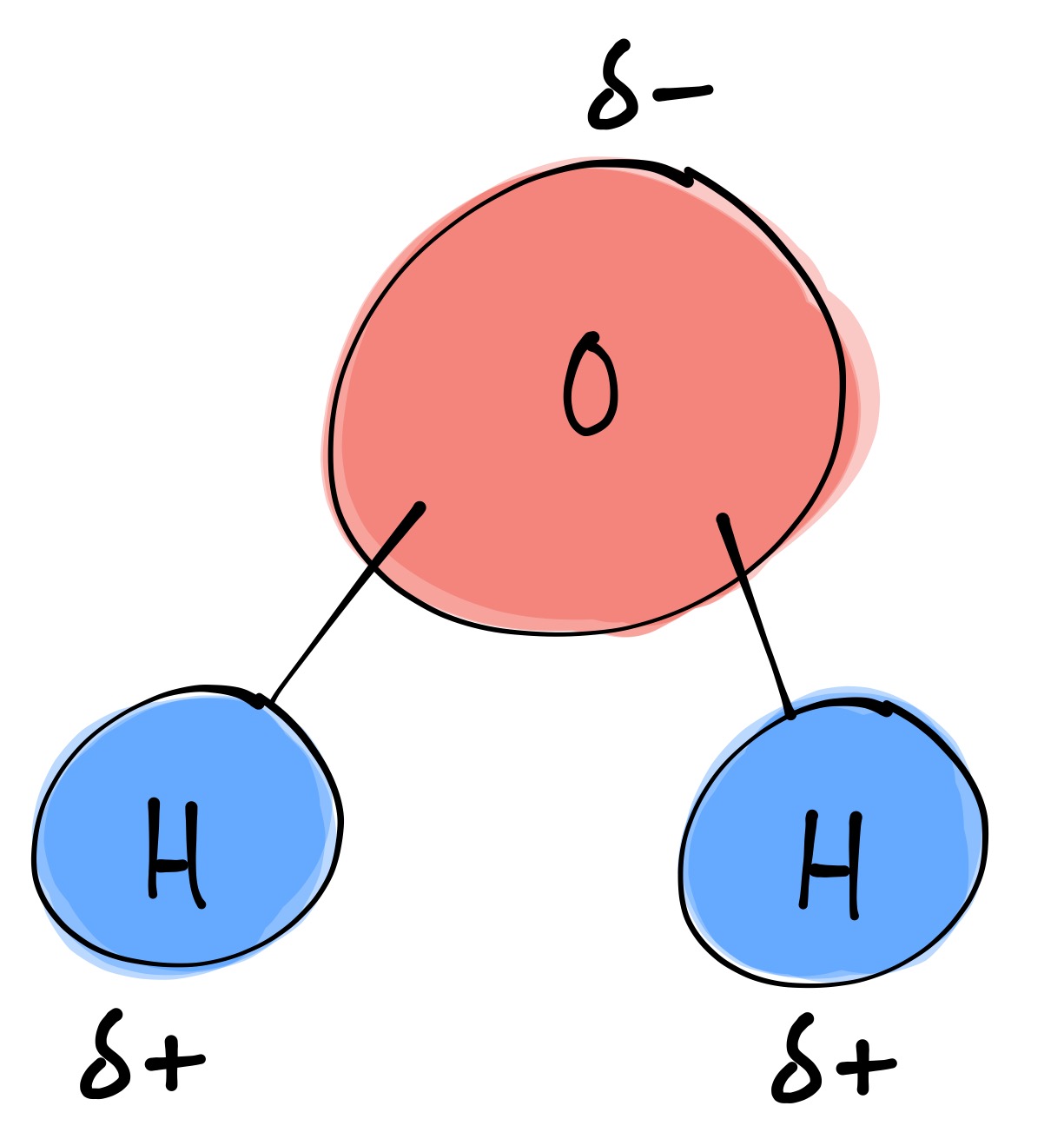
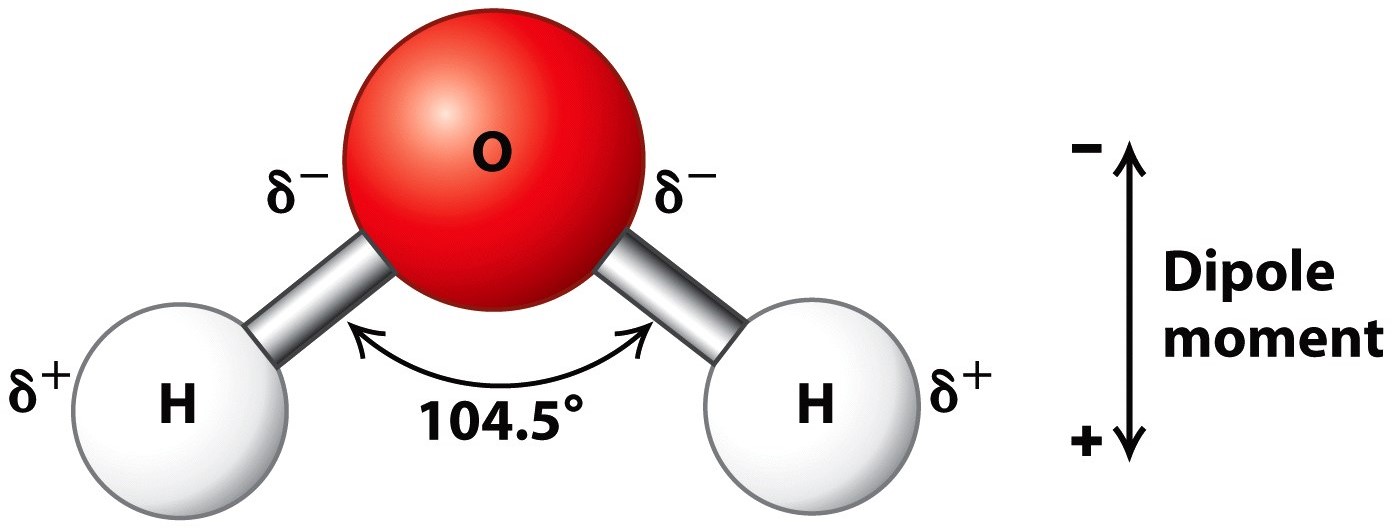
Draw A Water Molecule And Label The Partial Charges - The bent shape of the water molecule is critical because the polar o−h o − h bonds do not cancel one another and the molecule as a whole is polar. The water molecule, visualized three different ways: Web the partial negative charge on the o of one molecule can form a hydrogen bond with the partial positive charge on the hydrogens of other molecules. Web water's large dipole moment leads to hydrogen bonding. The h 2 o molecule is electrically neutral, but the positive and negative charges are not distributed uniformly. The electron cloud model shows where electrons are in a molecule. Web draw a sketch of two water molecules connected by a hydrogen bond. Video answer solved by verified expert video by farhan anwar university of arizona | answered on 02/28/2023 video answers to similar questions Web element that has a slight negative charge when sharing electrons within a water molecule. Web let u= {w, o, m, e, n}, m= {n, o, w}, and t= {w, o, n}. Arrange them and label where hydrogen bonding is occurring. There are two lone pairs of electrons on each oxygen atom (represented by. Use a dotted line to represent the hydrogen bond. The electron cloud model shows where electrons are in a molecule. The electronic (negative) charge is concentrated at the oxygen end of the molecule, owing. The electronic (negative) charge is concentrated at the oxygen end of the molecule, owing. Web the water molecule has a total of 10 protons and 10 electrons (8 from the oxygen atom and 1 from each of the two hydrogen atoms). This is illustrated by the gradation in color in the schematic diagram here. Arrange them and label where hydrogen. Water is polar due to the uneven sharing of electrons between oxygen and hydrogen. Web label charges and draw stick model that represents molecule; There are two lone pairs of electrons on each oxygen atom (represented by. Web water's large dipole moment leads to hydrogen bonding. Thus, in an aqueous environment, water molecules, which are at a very high. Label the bond between hydrogen and oxygen as polar covalent bond. The diagram below depicts a water molecule. Start studying label water molecule. The adjacent water molecules align themselves so that the partial charges on adjacen molecules can form an attraction. Complete the labels showing the locations of the hydrogen atoms, the oxygen atom, and the regions of. Web draw a water molecule with charges. Since it has the same number of protons and electrons, the water molecule is neutral. Sign up and see the remaining cards. Web the water molecule has a total of 10 protons and 10 electrons (8 from the oxygen atom and 1 from each of the two hydrogen atoms). The adjacent water molecules. Web partial positive charge and the oxygen atom acquires a partial negative charge. The diagram below depicts a water molecule. 1) indicate any full or partial charges, 2) label as symmetrical or asymmetrical, and 3) label as nonpolar or polar. The partial negative charge at one end of a water molecule is attracted to the partial positive charge of another. Web polar molecules have full or partial charges. Video answer solved by verified expert video by farhan anwar university of arizona | answered on 02/28/2023 video answers to similar questions Web water's large dipole moment leads to hydrogen bonding. Web partial positive charge and the oxygen atom acquires a partial negative charge. Partial charges are created due to the asymmetric. Label each atom and each location of a partial charge. Web water's large dipole moment leads to hydrogen bonding. Web water (h2o) should be drawn as two hydrogen atoms connected to one oxygen atom by a bond known as a polar covalent bond. Since it has the same number of protons and electrons, the water molecule is neutral. This is. Arrange them and label where hydrogen bonding is occurring. Water molecules are also attracted to other polar molecules and to ions. Web label charges and draw stick model that represents molecule; Web partial positive charge and the oxygen atom acquires a partial negative charge. Web the partial negative charge on the o of one molecule can form a hydrogen bond. Partial charges are created due to the asymmetric distribution of electrons in chemical bonds. Because the water molecule has an h — o — h bond angle of 105°, the molecule as a whole is polar. It is represented by the greek lowercase delta (𝛿), namely 𝛿− or 𝛿+. Draw a ring around an atom or a group of atoms. Sign up and see the remaining cards. (use flat bohr atoms showing the sharing of electrons in molecules.) electronegativity values are o=3.44, c=2.5, and h=2.1. Arrange them and label where hydrogen bonding is occurring. Web water's large dipole moment leads to hydrogen bonding. The bent shape of the water molecule is critical because the polar o−h o − h bonds do not cancel one another and the molecule as a whole is polar. Web element that has a slight negative charge when sharing electrons within a water molecule. This creates a partial charge called a dipole. Label the bond between hydrogen and oxygen as polar covalent bond. Learn vocabulary, terms, and more with flashcards, games, and other study tools. Web polar molecules have full or partial charges. Web water (h2o) should be drawn as two hydrogen atoms connected to one oxygen atom by a bond known as a polar covalent bond. Two water molecules attached to each other by hydrogen bond label hydrogen bonds this problem has been solved! The electronic (negative) charge is concentrated at the oxygen end of the molecule, owing. The partial negative charge of one water molecule is attracted to the partial positive charge of another water molecule. Water is polar due to the uneven sharing of electrons between oxygen and hydrogen. Web partial positive charge and the oxygen atom acquires a partial negative charge.
Types of Atoms Science at Your Doorstep

illustration of biochemistry, Water molecule consists of two hydrogen
The BioLogs CAPE 1 Water Introduction to it's BIOchemistry
Water Molecule Gay Eat Ass

The Structure and Properties of Water / Introduction to Chemistry
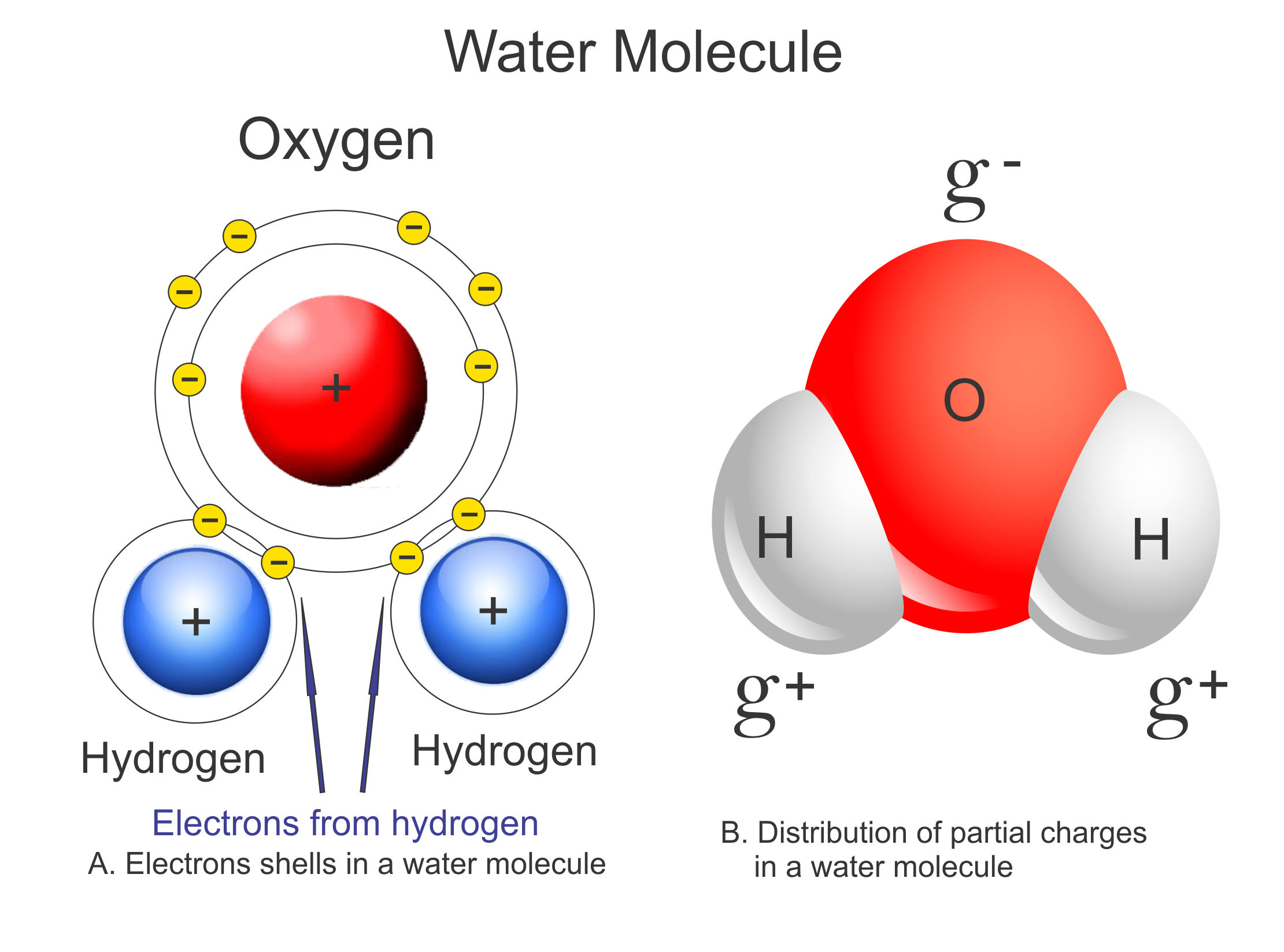
Polar Water Molecule Diagram
Describe the Structure of a Water Molecule

Water Molecule Structure For Kids

Water has both a hydrogen bond and a polar covalent bond. Hydrogen
Solved Label the following diagram of water molecules,
Web The Water Molecule Has A Total Of 10 Protons And 10 Electrons (8 From The Oxygen Atom And 1 From Each Of The Two Hydrogen Atoms).
Draw Water Vs Methane Molecules Showing The Electron Configurations.
Start Studying Label Water Molecule.
Web Draw A Sketch Of Two Water Molecules Connected By A Hydrogen Bond.
Related Post: