Draw All Resonance Structures For The Ozone Molecule O3
Draw All Resonance Structures For The Ozone Molecule O3 - Web the lewis structure of ozone has: If there are equivalent resonance structures, draw all of them. Solution verified by toppr (i) resonating structure of ozone ( o 3 ): Web i quickly take you through how to draw the lewis structure of o3 (ozone). In ozone, a molecular orbital extending over all three oxygen atoms is formed from three atom centered pz orbitals. Draw lewis structure(s) for the ozone molecule (o3). (figure 1) (ii) resonating structure of n o 3 −: Both bonds in ozone have a bond order of 1.5 and are the same length, which is longer than a double bond (because the double bond is a stronger bond) and shorter than a single bond (because a single bond is a weaker bond). Do not draw resonance arrows between your structures. * two resonance structures * a lone pair on the central oxygen atom * ax2e vsepr. Web chemistry bond parameters question draw the resonating structure of (i) ozone ( o 3) molecule (ii)nitrate ion ( n o 3 − ). Do not draw resonance arrows between your structures. Each step of drawing the lewis structure of o 3 is explained in detail in this tutorial. Web lewis structure of o 3 can be drawn by starting. The molecule’s 1/2 bond order, which indicates the number of bonds between two atoms, also supports the notion that the hybrid structure incorporates the ozone molecule’s two principal resonance configurations. Web ozone molecules are three oxygen atoms bonded in succession, they are not in a ring.the middle oxygen, according to the lewis structure, needs one double bon. If we draw. Web science chemistry chemistry questions and answers draw all resonance structures for the ozone molecule, o3. Both bonds in ozone have a bond order of 1.5 and are the same length, which is longer than a double bond (because the double bond is a stronger bond) and shorter than a single bond (because a single bond is a weaker bond).. Web figure 5.3.4 the resonance structure of ozone involves a molecular orbital extending all three oxygen atoms. * two resonance structures * a lone pair on the central oxygen atom * ax2e vsepr. The two possible lewis structures that can be drawn for ozone are I also go over the resonance, hybridization, shape and bond angle. Draw all resonance structures. Include all lone pairs and/or radicals. Web science chemistry chemistry questions and answers draw all resonance structures for the ozone molecule, o3. Web draw the most valid lewis structure for ozone (o3): Do not draw resonance arrows between your structures. Draw all resonance structures for the ozone molecule, o3. Web i quickly take you through how to draw the lewis structure of o3 (ozone). If there are equivalent resonance structures, draw all of them. The two possible lewis structures that can be drawn for ozone are In ozone, a molecular orbital extending over all three oxygen atoms is formed from three atom centered pz orbitals. Web ozone molecules are. Be sure to include all resonance structures that satisfy the octet rule. Draw lewis structure(s) for the ozone molecule (o3). Draw the lewis structure for the ozone (03) molecule. * three oxygen atoms in a row. (figure 2) was this answer helpful? For molecules with multiple resonance structures, draw all possible resonance structures (in the same drawing window): Draw lewis structure(s) for the ozone molecule (o3). Both bonds in ozone have a bond order of 1.5 and are the same length, which is longer than a double bond (because the double bond is a stronger bond) and shorter than a single bond. Do not show ion charges in your drawings. Each step of drawing the lewis structure of o 3 is explained in detail in this tutorial. Do not draw resonance arrows between your structures. In ozone, a molecular orbital extending over all three oxygen atoms is formed from three atom centered pz orbitals. Draw one structure per sketcher box, and separate. Draw lewis structure(s) for the ozone molecule (o3). If there are equivalent resonance structures, draw all of them. Both bonds in ozone have a bond order of 1.5 and are the same length, which is longer than a double bond (because the double bond is a stronger bond) and shorter than a single bond (because a single bond is a. Web lewis structure of o 3 can be drawn by starting from valence electrons of oxygen atoms in several steps. The molecule’s 1/2 bond order, which indicates the number of bonds between two atoms, also supports the notion that the hybrid structure incorporates the ozone molecule’s two principal resonance configurations. Web for example, drawing one lewis structure for ozone (o 3) gives us a misleading picture of the actual bonding in the molecule. Web i quickly take you through how to draw the lewis structure of o3 (ozone). Be sure to include all resonance structures that satisfy the octet rule. Similar molecular orbitals are found in every resonance structure. Press and hold explanation check. * three oxygen atoms in a row. Draw one structure per sketcher box, and separate added sketcher boxes with the ↔ symbol. Do not show ion charges in your drawings. Web figure 5.3.4 the resonance structure of ozone involves a molecular orbital extending all three oxygen atoms. Web draw the most valid lewis structure for ozone (o3): In ozone, a molecular orbital extending over all three oxygen atoms is formed from three atom centered pz orbitals. Both bonds in ozone have a bond order of 1.5 and are the same length, which is longer than a double bond (because the double bond is a stronger bond) and shorter than a single bond (because a single bond is a weaker bond). (figure 1) (ii) resonating structure of n o 3 −: Solution verified by toppr (i) resonating structure of ozone ( o 3 ):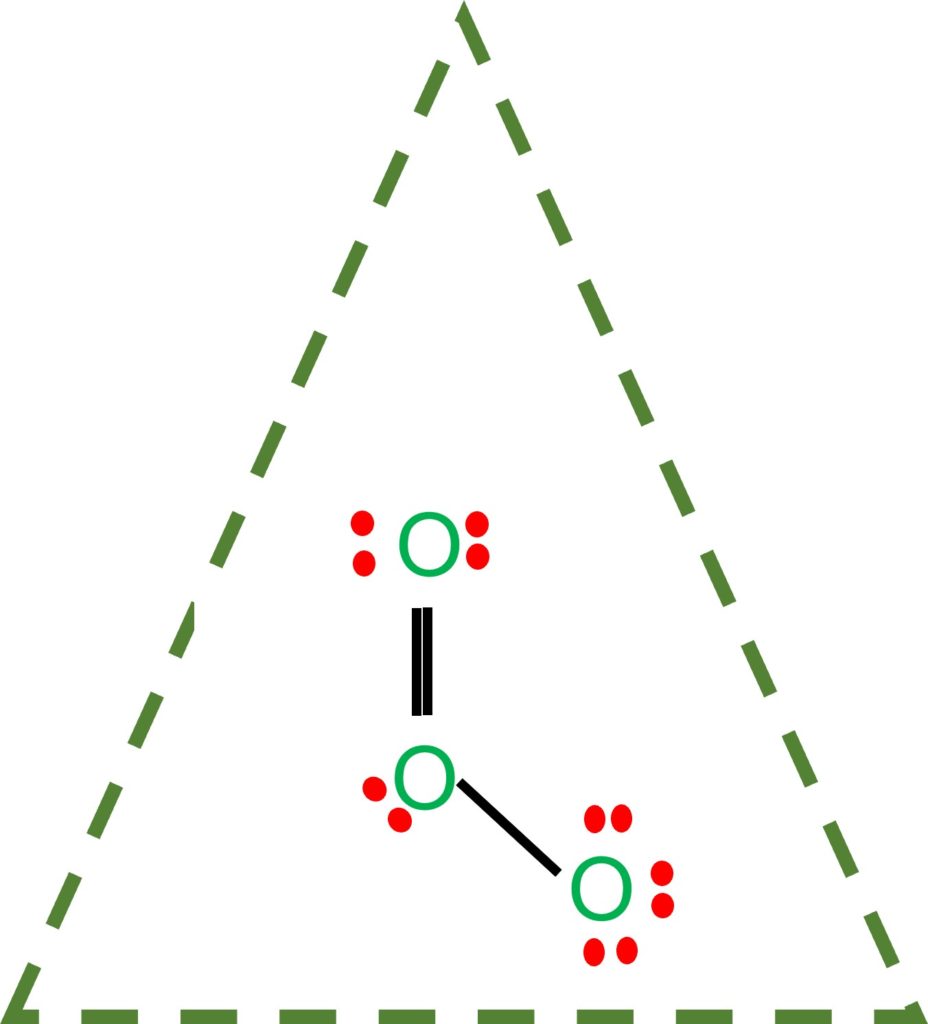
O3 Lewis Structure Step By Step Drawing What's Insight
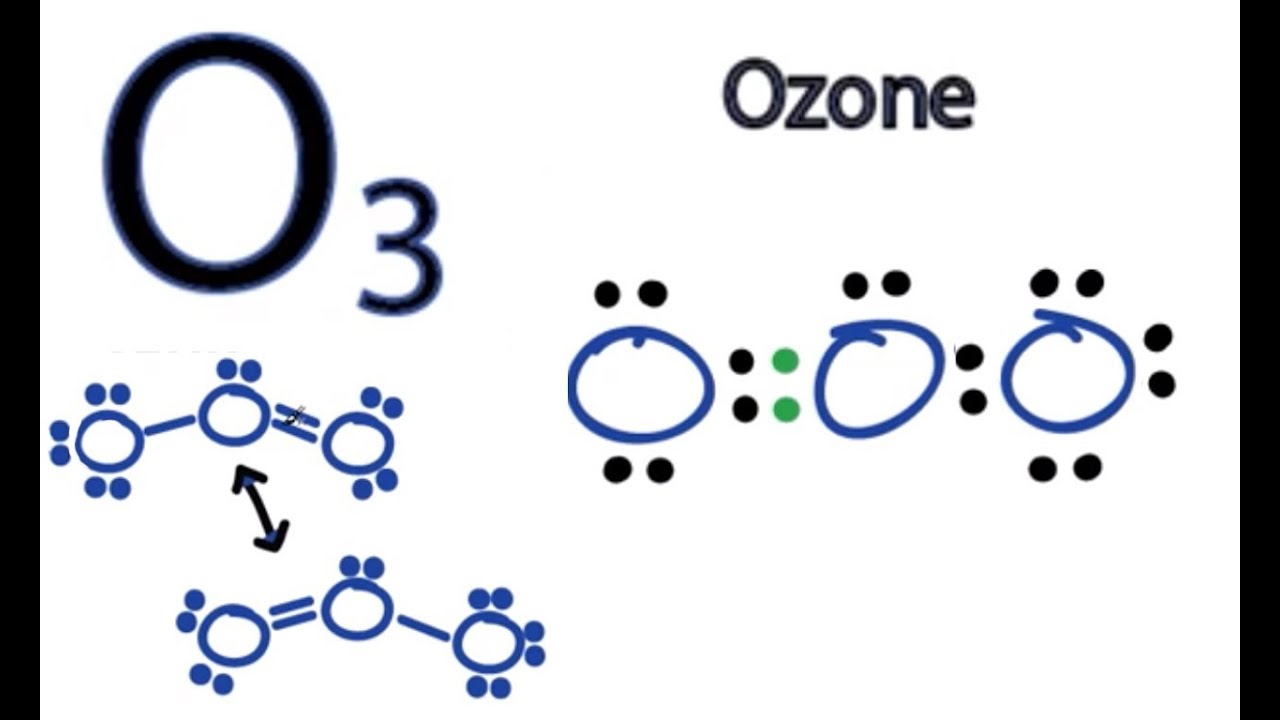
O3 Lewis Structure How to Draw the Dot Structure for O3 YouTube
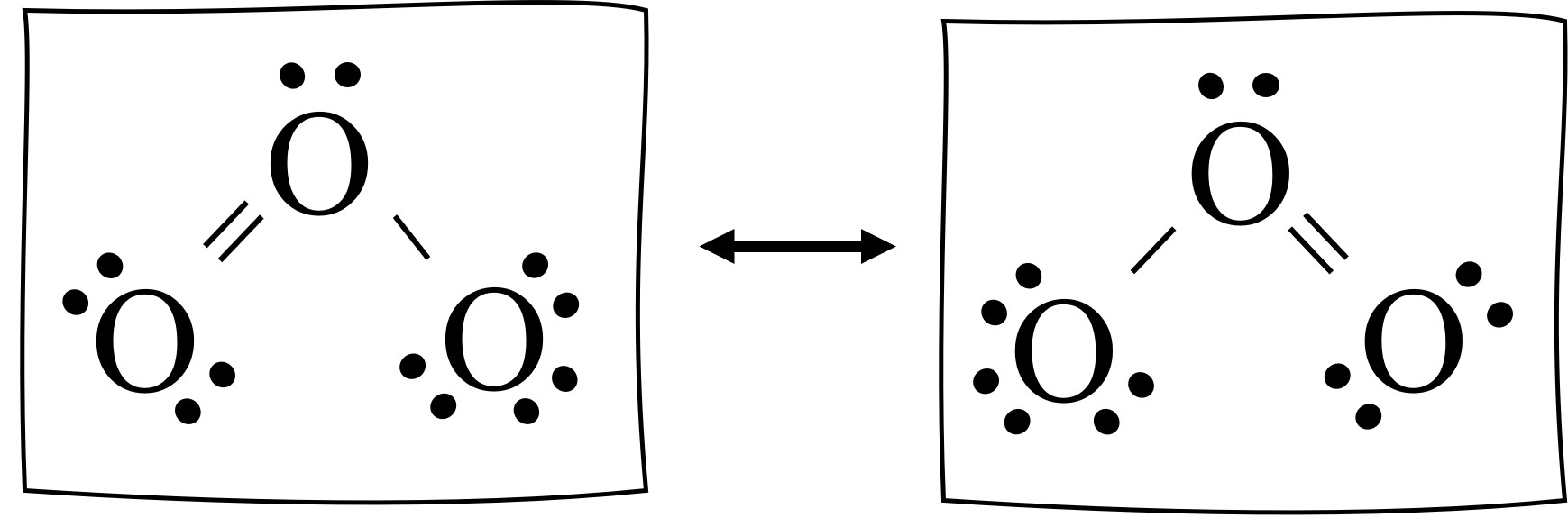
Resonance Structures Easy Hard Science

12+ O3 Lewis Structure Robhosking Diagram

O3 Lewis Structure (Ozone) Chemistry, Ozone, Lewis
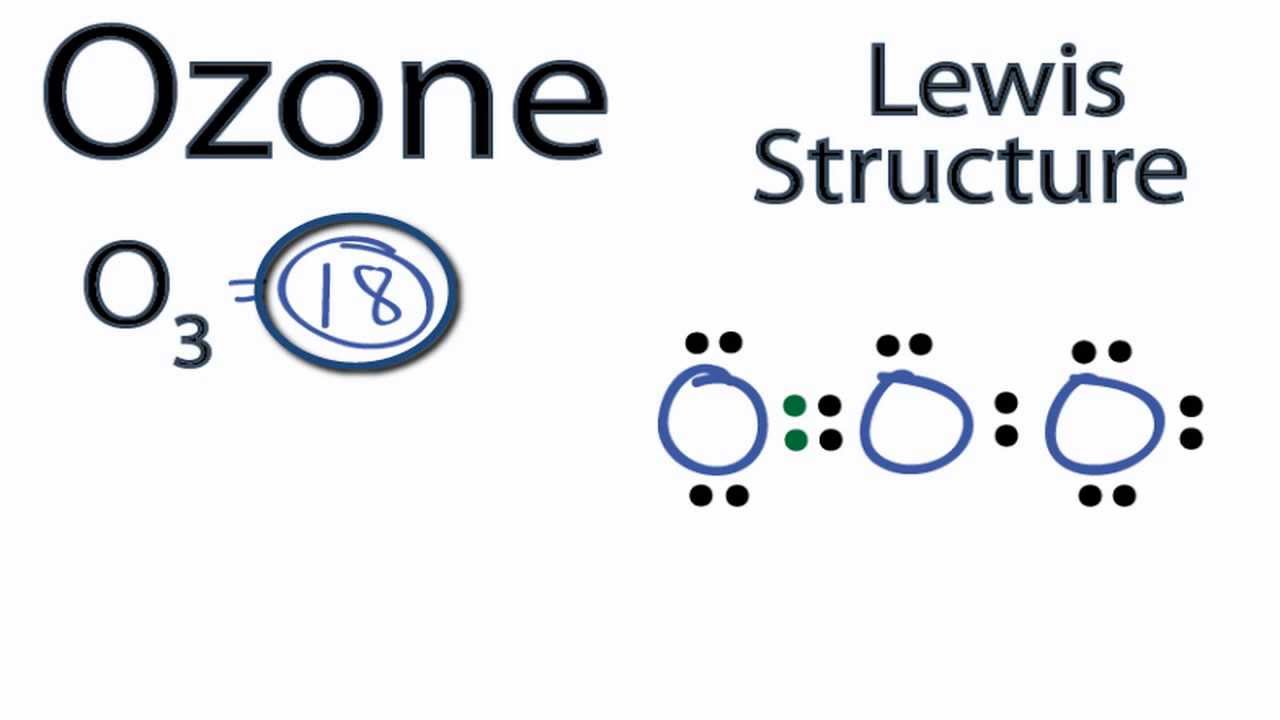
Ozone Lewis Structure How to Draw the Lewis Structure for Ozone YouTube
Resonance Presentation Chemistry
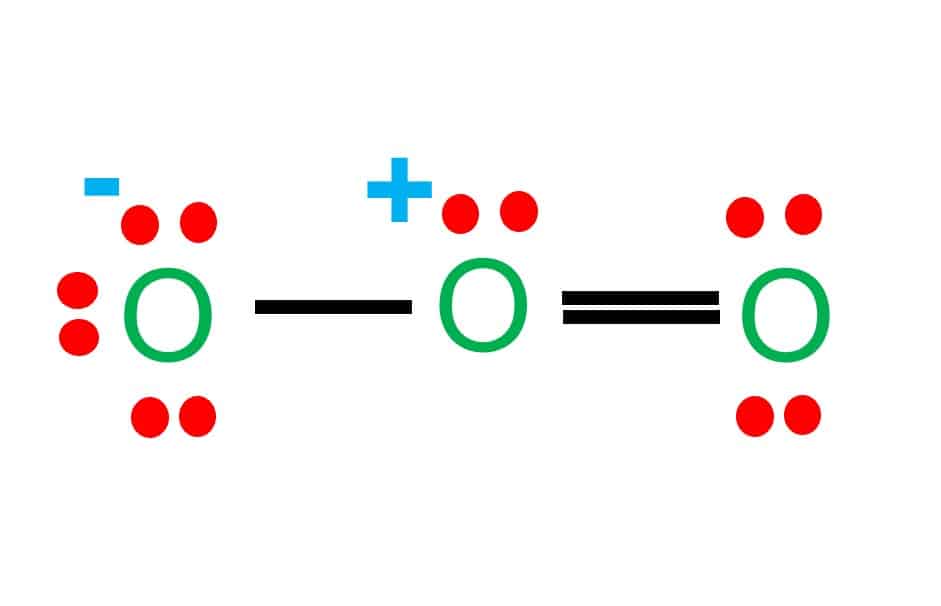
O3 Lewis Structure Step By Step Drawing What's Insight

Resonance Structures of O3, Ozone YouTube

O3 Resonance Structures (Ozone) YouTube
Draw Lewis Structure(S) For The Ozone Molecule (O3).
Include All Lone Pairs And/Or Radicals.
If There Are Equivalent Resonance Structures, Draw All Of Them.
It's An Average Of Them.
Related Post:
.PNG)