Draw As Many Unique Lewis Structures As Possible For C4H8
Draw As Many Unique Lewis Structures As Possible For C4H8 - Butene in your notebook, draw as many unique lewis structures for c4he as possible. Web in chemistry, isomers are molecules with identical molecular formulas — that is, the same number of atoms of each element — but distinct arrangements of atom. Web the possible lewis structures of c₄h₈ with every lewis structure include one double bond between 2c atoms is attached as a picture. Web draw as many unique lewis structures as possible for butane, c4h8 question 1: Web representing a covalent bond using lewis structures. This hydrocarbon has two hydrogens less, so it must contain either a double bond or a ring. You do not need to draw those structures or make those models. The alkene butene, often known as butylene, has the chemical formula c₄h₈. Web draw as many unique lewis structures as possible for c4h8. Any of the several chemicals may be referred to by the word butene. Drawing lewis structures for molecules with one central atom: Web chemistry chemistry questions and answers art 4. #1 draw a rough skeleton structure #2 mention lone pairs on the atoms #3 if needed, mention formal charges on the atoms #4 minimize formal charges by converting lone pairs of the atoms, and try to get a stable lewis structure #5 repeat. The lewis diagram for n 2 is as follows: Determine the total number of valence (outer shell) electrons in the molecule or ion. Every lewis structure should include one double bond between 2 c atoms. #1 draw a rough skeleton structure #2 mention lone pairs on the atoms #3 if needed, mention formal charges on the atoms #4 minimize formal. Web representing a covalent bond using lewis structures. Draw and upload a lewis structure. 2 4 3 1 5 or more question 2: Determine the total number of valence (outer shell) electrons in the molecule or ion. The total number of electrons is 2 x 5 = 10 electrons. This hydrocarbon has two hydrogens less, so it must contain either a double bond or a ring. Draw a lewis structure c4h8 draw a lewis structure c4h8 here’s the best way to solve it. There are two possible structures that form a ring. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. The. The sum of the valence electrons is. The top one in pic trans isomer with hydrogen on opposite side and double bond in middle cis isomer with hydrogens on same side with double bond in the middle shorten carbon chain by one and put it in the middle put carbons in a ring and form a box make 3 carbon. In ch 2 o, the central atom is surrounded by two different types of atoms. Web draw as many unique lewis structures as possible for butane, c4h8 question 1: How many unique structures could you make? Every lewis structure should include one double bond between two c atoms. The alkene butene, often known as butylene, has the chemical formula c₄h₈. The total number of electrons is 4 x 2 (1) + 6 = 12 electrons. Isomers of c4h8 with double bond. Draw and upload a lewis structure. Web draw as many lewis structures of c4h8 as possible. Web draw as many unique lewis structures as possible for butane, c4h8 question 1: I count six isomers of c_4h_8. Web representing a covalent bond using lewis structures. Determine the total number of valence (outer shell) electrons. There is more than one acceptable diagram with the same answers. For very simple molecules and molecular ions, we can write the lewis structures by merely pairing up the unpaired electrons on the constituent atoms. (b) for each structure, show how many types of h would appear in the proton nmr spectrum. Every lewis structure should include one double bond between two c atoms. The bond between the two nitrogen atoms is a triple bond. How many unique structures could you make? Determine the total number of valence (outer shell) electrons. (c) for each structure, show how many types of c would appear in the 13c nmr spectrum. Every lewis structure should include one double bond between two c atoms. Web expert solution trending now this is a popular solution! C4h8 with a double bond, also known as butene isomers, are organic compounds that contain four carbon atoms and eight hydrogen. The total number of electrons is 2 x 5 = 10 electrons. (d) if an unknown compound of formula c4h8 shows two types of h and three. > an alkane with 4 carbon atoms would have the formula c_4h_10. The lewis diagram for n 2 is as follows: Web here’s how you can easily draw the c 4 h 8 lewis structure step by step: (b) for each structure, show how many types of h would appear in the proton nmr spectrum. There are 2 steps to solve this one. Physical and chemical properties of cis and trans structures are different. Isomers of c4h8 with double bond. Web in c 4 h 8, we can draw two geometrical isomers as cis and trans. Draw and upload a lewis structure. Do not consider cyclic (ring) structures. Determine the total number of valence (outer shell) electrons. Any of the several chemicals may be referred to by the word butene. Web draw lewis structures for molecules. I count six isomers of c_4h_8.33. Structural isomers possible for c4h8br2 are (1) 3 (2) 9 (3) 10 (4) 4
C4h8 Lewis Structure Isomers
Butene C4H8 Molecular Geometry Hybridization Molecular Weight
[Solved] Referring to the isomers of C4H8, can Molecule 1 be made to

C4H8 Lewis Structure How to Draw the Lewis Structure for C4H8 YouTube

Solved A Student Drew The Following Six Structures For C4...

Draw all isomers for C4H8 YouTube

Draw structures for all constitutional isomers with the molecular
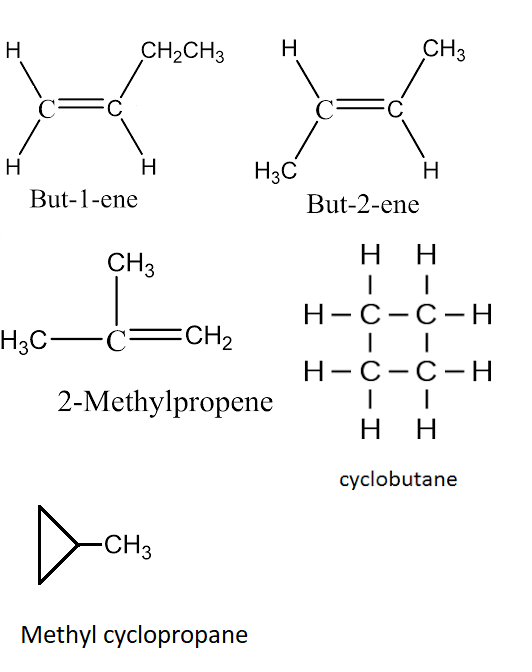
C4h8 Lewis Structure Isomers
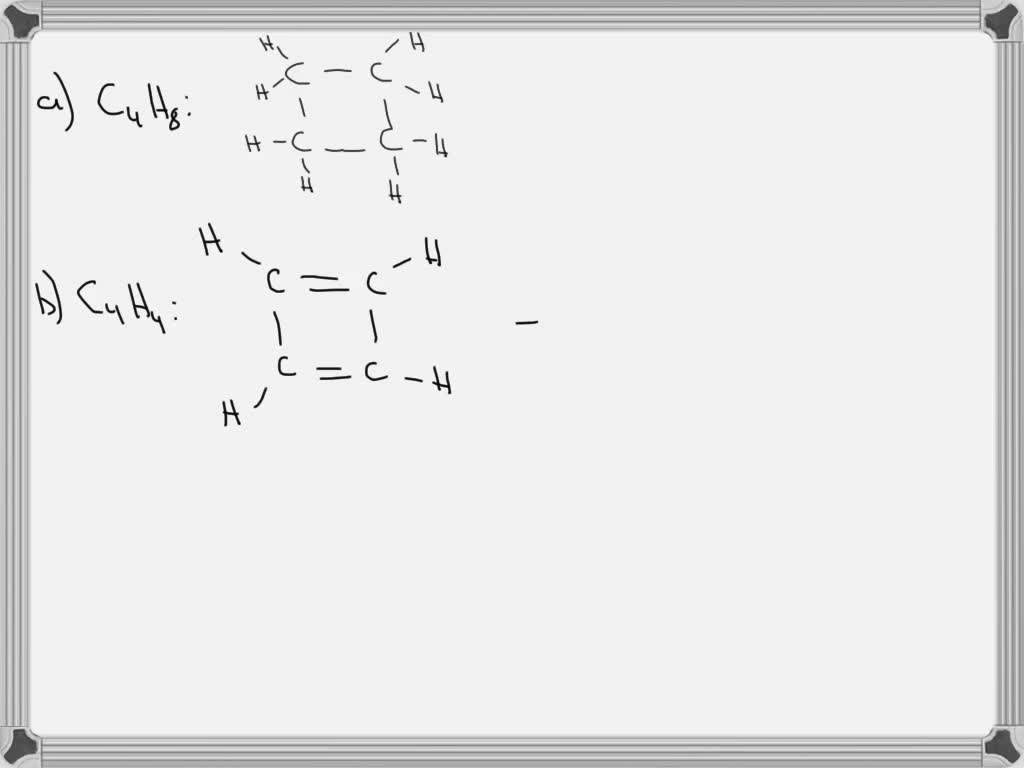
SOLVED Carbon ring structures are common in organic chemistry. Draw a
Use Your Diagram To Answer The Following Questions.
This Hydrocarbon Has Two Hydrogens Less, So It Must Contain Either A Double Bond Or A Ring.
Web Draw As Many Unique Lewis Structures As Possible For C4H8.
The Total Number Of Electrons Is 4 X 2 (1) + 6 = 12 Electrons.
Related Post:

