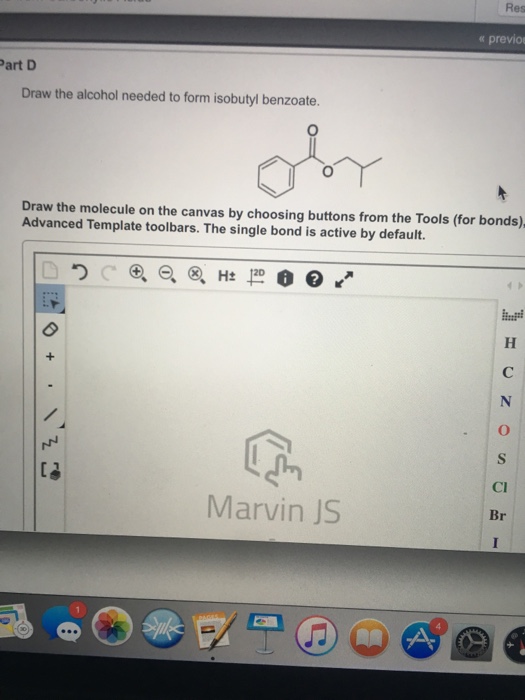
Draw The Alcohol Needed To Form Isobutyl Benzoate
Draw The Alcohol Needed To Form Isobutyl Benzoate - Web an ester is an organic compound that is a derivative of a carboxylic acid in which the hydrogen atom of the hydroxyl group has been replaced with an alkyl group. Draw the molecule on the canvas by choosing buttons from the tools (for bonds), atoms, and advanced template toolbars. Draw the molecule on the canvas by choosing buttons from the tools (for bonds), atoms, and advanced template toolbars. If an acid chloride undergoes an addition reaction with an organometallic reagent, what type of… This substance is the reference (100 level) for octane ratings. One way to make an ester is to use a fischer esterification reaction. The alcohol needed is the isobutanol. Constants periodic table draw the alcohol needed to form isobutyl benzoate. It has a role as a metabolite. To form isobutyl benzoate you need to react isobutanol with benzoic acid in a reaction called esterification of carboxylic acids with alcohols, in which water is also formed. A chemical reaction between an alcohol and an acid, in which an ester is formed. The alcohol needed is the isobutanol. The reaction between isobutanol and benzoic acid will produce isobutyl benzoate, with water as a byproduct. The single bond is active. To form isobutyl benzoate you need to react isobutanol with benzoic acid in a reaction called esterification of. We don’t have your requested question, but here is a suggested video that might help. Dry hydrogen chloride gas is used in some cases, but these tend to involve aromatic esters (ones containing a benzene ring). Here’s the best way to solve it. To form isobutyl benzoate you need to react isobutanol with benzoic acid in a reaction called esterification. Dry hydrogen chloride gas is used in some cases, but these tend to involve aromatic esters (ones containing a benzene ring). We need an alcohol to form an ester (isobutyl benzoate). Draw the molecule on the canvas by choosing buttons from the tools (for bonds) advanced template toolbars. Here’s the best way to solve it. Web expert answer 100% (3. Draw the structure of a. To form isobutyl benzoate you need to react isobutanol with benzoic acid in a reaction called esterification of carboxylic acids with alcohols, in which water is also formed. View available hint (s) oh. Draw the molecule on the canvas by choosing buttons from the tools (for bonds), atoms, and advanced template toolbars. You'll get a. We don’t have your requested question, but here is a suggested video that might help. It is functionally related to a benzoic acid and an isobutanol. B) write the names of the carboxylic acid and alcohol underneath your drawings. Isobutyl benzoate is a benzoate ester which is obtained by the condensation of benzoic acid and… q: Isobutyl benzoate is an. To form isobutyl benzoate you need to react isobutanol with benzoic acid in a reaction called esterification of carboxylic acids with alcohols, in which water is also formed. Draw the structural formula for each of the following. Dry hydrogen chloride gas is used in some cases, but these tend to involve aromatic esters (ones containing a benzene ring). Draw the. B) write the names of the carboxylic acid and alcohol underneath your drawings. The catalyst is usually concentrated sulphuric acid. Web 178.23 g/mol computed by pubchem 2.2 (pubchem release 2021.10.14) dates create: Web an ester is an organic compound that is a derivative of a carboxylic acid in which the hydrogen atom of the hydroxyl group has been replaced with. So if you start with the carboxylic acid, and you add an alcohol, and a source of protons, you're gonna form your ester, and you're also going to make water in this process. Isobutyl benzoate is an ester and so an carboxylic acid and an alcohol are needed to form isobutyl benzoate. Web esters are produced when carboxylic acids are. It is functionally related to a benzoic acid and an isobutanol. The reaction can be written as follows: A chemical reaction between an alcohol and an acid, in which an ester is formed. One way to make an ester is to use a fischer esterification reaction. Here’s the best way to solve it. Identify the following as a fatty acid, soap, triacylglycerol, wax, glycerophospholipid, sphingolipid, or steroid: Draw the molecule on the canvas by choosing buttons from the tools (for bonds), atoms, and advanced template toolbars. Web 178.23 g/mol computed by pubchem 2.2 (pubchem release 2021.10.14) dates create: Dry hydrogen chloride gas is used in some cases, but these tend to involve aromatic. Identify the following as a fatty acid, soap, triacylglycerol, wax, glycerophospholipid, sphingolipid, or steroid: Isobutyl benzoate is a benzoate ester which is obtained by the condensation of benzoic acid and… q: Draw the structural formula for each of the following. Isobutyl benzoate is an ester and so an carboxylic acid and an alcohol are needed to form isobutyl benzoate. The alcohol needed is the isobutanol. To understand how esters are formed from carboxylic acids and alcohols when carboxylic acids and alcohols are heated in the presence of an acid catalyst, they will react to form an ester and water. Draw the molecule on the canvas by choosing buttons from the tools (for bonds), atoms, and advanced template toolbars. To form isobutyl benzoate you need to react isobutanol with benzoic acid in a reaction called esterification of carboxylic acids with alcohols, in which water is also formed. We don’t have your requested question, but here is a suggested video that might help. Draw the molecule on the canvas by choosing buttons from the tools (for bonds) advanced template toolbars. View available hint (s) oh. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. The single bond is active. Web 178.23 g/mol computed by pubchem 2.2 (pubchem release 2021.10.14) dates create: Web this problem has been solved! We need an alcohol to form an ester (isobutyl benzoate).Solved Draw the alcohol needed to form isobutyl benzoate.

Isobutyl benzoate SIELC

draw the alcohol needed to form isobutyl benzoate

draw the alcohol needed to form isobutyl benzoate
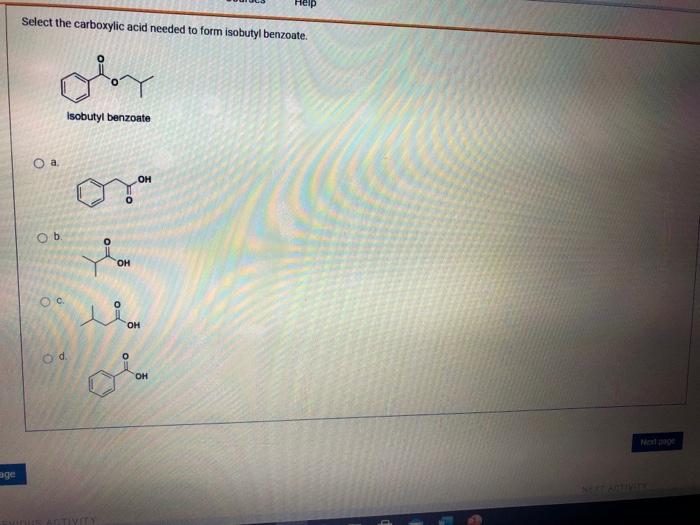
Solved Select the carboxylic acid needed to form isobutyl

Which of the following alkenes when passed through conc. H2SO4 followed
Solved Draw The Water That Is Formed From The Reaction Of...
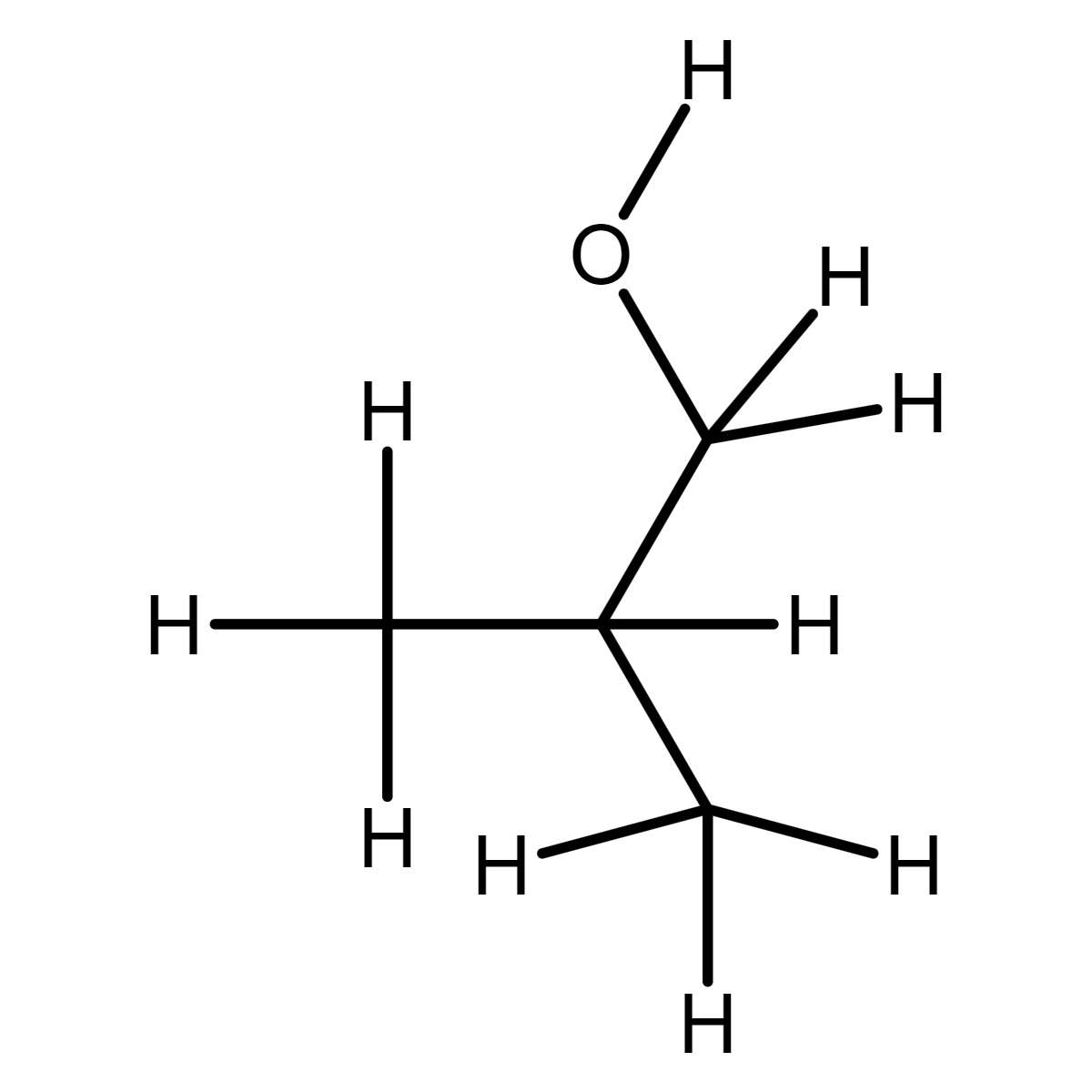
Isobutyl alcohol solution CRM LABSTANDARD

What is the difference between isobutyl and sec butyl?
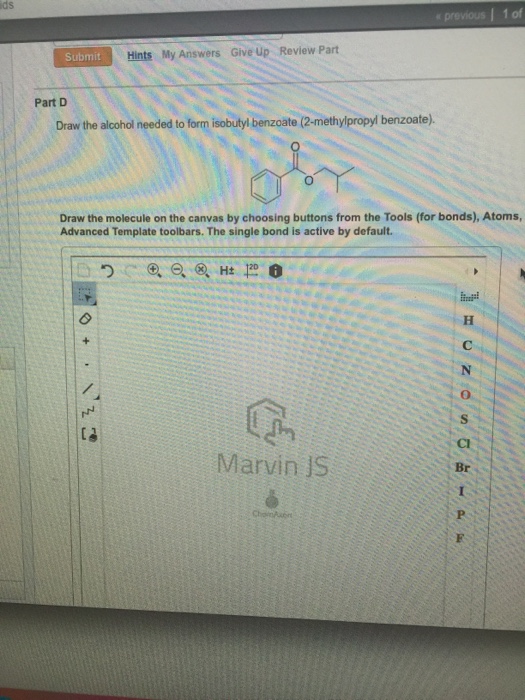
[Solved] Draw the alcohol needed to form isobutyl benzoate (2
Draw The Molecule On The Canvas By Choosing Buttons From The Tools (For Bonds), Atoms, And Advanced Template Toolbars.
Here’s The Best Way To Solve It.
They Are Shown In The Attached File.
This Substance Is The Reference (100 Level) For Octane Ratings.
Related Post: