Draw The Electron Configuration For A Neutral Atom Of Silicon
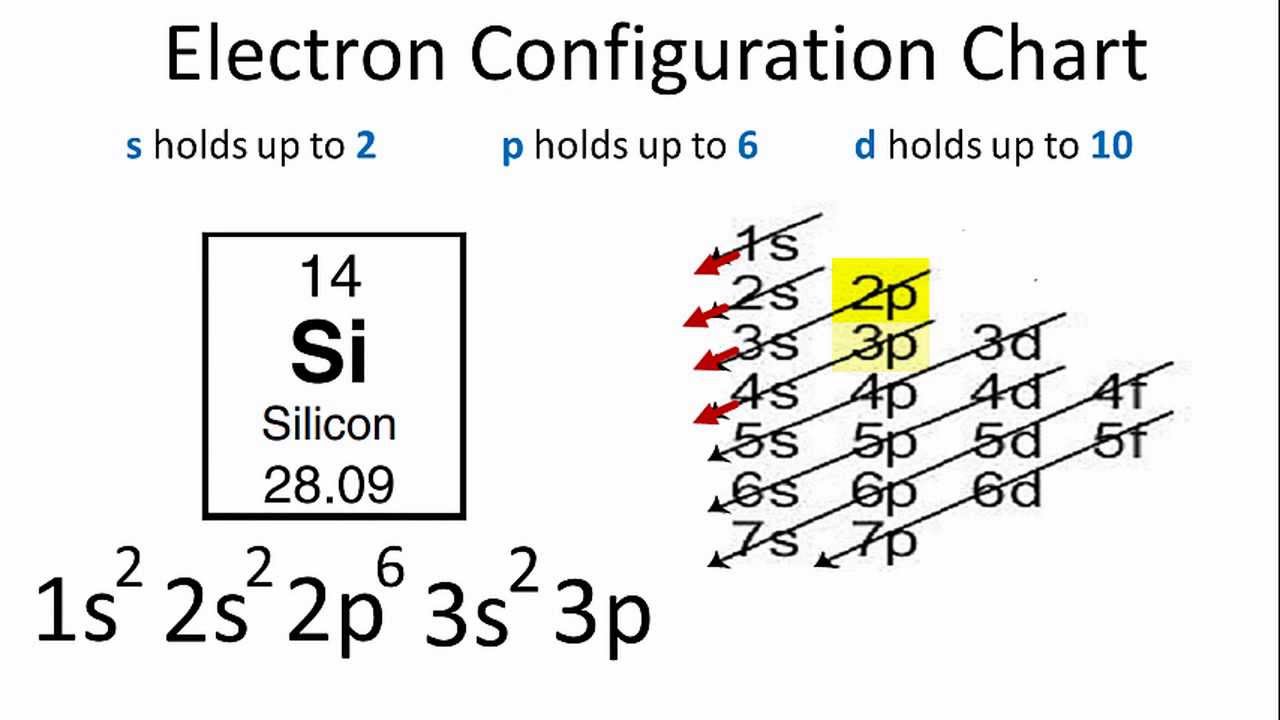
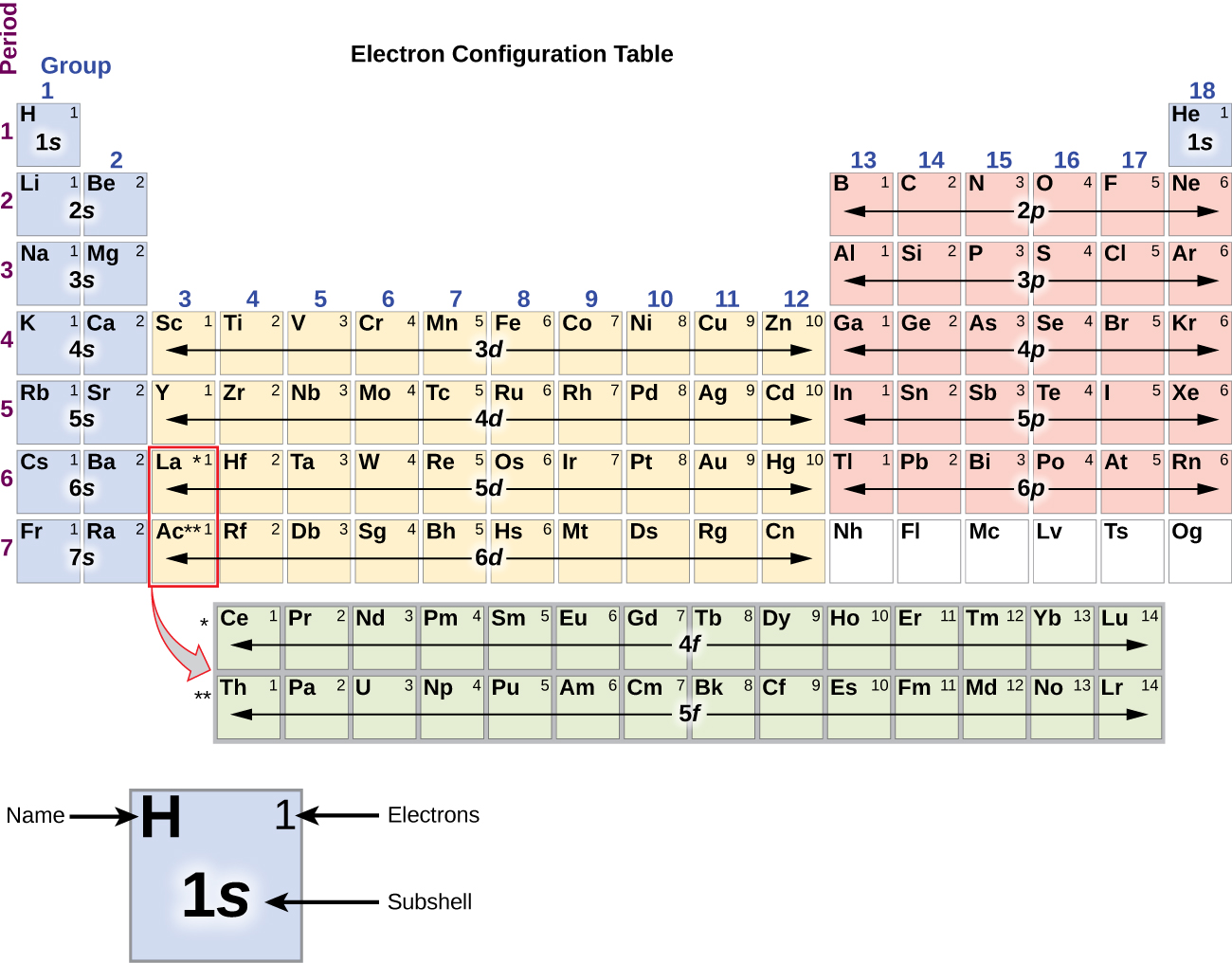
Draw The Electron Configuration For A Neutral Atom Of Silicon - The isotope is defined by the number of neutrons in an atom, which might be equal to the number of protons—or not. Remember, a neutral atom contains the same number of protons and electrons. Web the neutral atom chlorine (z=17), for instance has 17 electrons. It has four valance electrons like carbon. A neutral chlorine atom has 17 electrons. Determine how many electrons were lost. Web what is the electron configuration of a neutral chlorine atom? (2 electrons) so, the electron configuration for a neutral silicon atom is: The p, d, and f orbitals have different sublevels, thus can hold more electrons. You'll get a detailed solution from a. A neutral chlorine atom has 17 electrons. Web typically, you need at least 8 steps to determine the electron configuration, starting with finding the atomic number by looking at the list of orbitals and understanding the notation. Web an electrically neutral atom has the following electron configuration: Web the arrangement of electrons in silicon in specific rules in different orbits. Web using figure \(\pageindex{2}\) as your guide, write the electron configuration of a neutral chlorine atom. Si 4 + was formed by the loss of four electrons. Energy 1 1 х ? It has four valance electrons like carbon. The shorthand electron configuration (or noble gas configuration) as well as full electron configuration is also mentioned in the. The electron configuration of silicon is [ ne] 3s 2 3p 2 , if the electron arrangement is through orbitals. Web a silicon atom is a neutral atom that has an atomic number of 14 which implies it has a total of 14 electrons. Web the upper right side shows the number of electrons in a neutral atom. Remove electrons. Web the arrangement of electrons in silicon in specific rules in different orbits and orbitals is called the electron configuration of silicon. Web what is the electron configuration of a neutral chlorine atom? This problem has been solved! Web march 23, 2023 by jay electron configuration chart of all elements is mentioned in the table below. Silicon has an atomic. The outer valance electron configuration of # s^2 p^2 allows silicon to have common charges of +2 and +4. Therefore, its ground state electronic configuration can be written as 1s 2 2s 2 2p 6 3s 2 3p 5. What is the name of this atom? You'll get a detailed solution from a. Web march 23, 2023 by jay electron. Therefore, the number of electrons in neutral atom of silicon is 14. Web the neutral atom chlorine (z=17), for instance has 17 electrons. Two electrons can go into the 1s subshell, 2 can go into the 2s subshell, and 6 can go into the 2p subshell. This problem has been solved! Let's draw the electron configuration for a neon neon,. When we write the configuration we'll put all 14 electrons in orbitals around the nucleus of the silicon atom. Of those 7 electrons, 2 can go into the 3s subshell, and the remaining 5 electrons can go into the. Write the configuration of the neutral atom. The shorthand electron configuration (or noble gas configuration) as well as full electron configuration. 1s 2 2s 2 2p 6 3s 1 I show you where silicon is on the periodic table and how to determine how many valence electrons silicon has. Remove electrons from the highest shell, starting with the highest energy subshell. Write the configuration of the neutral atom. A neutral chlorine atom has 17 electrons. The shorthand electron configuration (or noble gas configuration) as well as full electron configuration is also mentioned in the. Silicon has an atomic number of 14, so a neutral silicon atom would have 14 electrons. Web the arrangement of electrons in silicon in specific rules in different orbits and orbitals is called the electron configuration of silicon. When we write. A neutral chlorine atom has 17 electrons. You'll get a detailed solution from a. As stated, the electron configuration of each element is unique to its position on the periodic table. Electron configuration can be done in two ways. When we write the configuration we'll put all 14 electrons in orbitals around the nucleus of the silicon atom. Web o electronic structure drawing a box diagram of the electron configuration of an atom draw the electron configuration for a neutral atom of silicon. Si 4 + was formed by the loss of four electrons. Energy 1 1 х ? An atom has a valence shell electron configuration of #ns^1#. As stated, the electron configuration of each element is unique to its position on the periodic table. Web using figure \(\pageindex{2}\) as your guide, write the electron configuration of a neutral chlorine atom. The shorthand electron configuration (or noble gas configuration) as well as full electron configuration is also mentioned in the. Determine how many electrons were lost. The atomic number of cl is 17. Write the configuration of the neutral atom. Web the neutral atom chlorine (z=17), for instance has 17 electrons. Therefore, its ground state electronic configuration can be written as 1s 2 2s 2 2p 6 3s 2 3p 5. 5 10 11 12 13 14 draw the electron configuration for a neutral atom of silicon energy x subu alignment continue giv Web the four different types of orbitals (s,p,d, and f) have different shapes, and one orbital can hold a maximum of two electrons. Web an electrically neutral atom has the following electron configuration: This problem has been solved!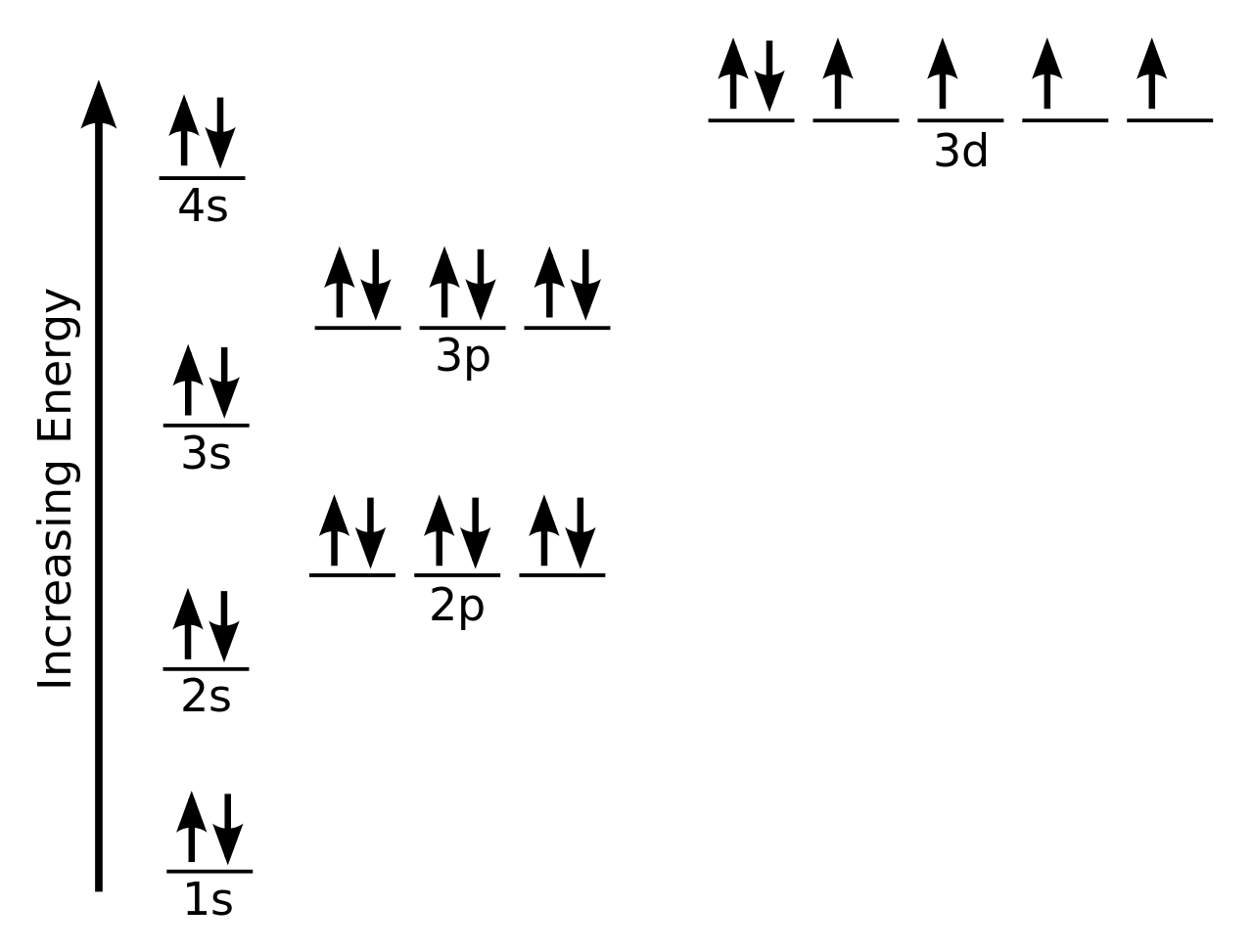
Silicon Electron Configuration (Si) with Orbital Diagram
![]()
Silicon Electron Configuration Orbital Diagram For Silicon (Si)
![]()
Silicon Atom Science Notes and Projects
![]()
Silicon Si (Element 14) of Periodic Table Elements FlashCards
Silicon Electron Configuration (Si) with Orbital Diagram
![]()
Symbol and electron diagram for silicon Royalty Free Vector
Silicon Electron Configuration How Many Unpaired Electrons Ellis
![]()
Electron arrangements

Electronic Configurations Intro Chemistry LibreTexts

Electron Configuration Chart With Orbitals
(2 Electrons) So, The Electron Configuration For A Neutral Silicon Atom Is:
Silicon Has An Atomic Number Of 14, So A Neutral Silicon Atom Would Have 14 Electrons.
When We Write The Configuration We'll Put All 14 Electrons In Orbitals Around The Nucleus Of The Silicon Atom.
But Wait — You Can Avoid All This If You Use Our Tool!
Related Post: