Draw The Lewis Dot Diagram For A Anion
Draw The Lewis Dot Diagram For A Anion - Exceptions ( 0) lewis dot structures: Write the 2 atomic symbols side by side. First, write the empirical formula of the compound down to see which elements are involved and how many atoms of each. Web draw lewis structures for ionic compounds. Web draw the lewis dot diagram for a h−anion. Likewise, they can be used to show the formation of anions from atoms, as shown here for chlorine and sulfur: Web a lewis diagram shows how the valence electrons are distributed around the atoms in a molecule. Web drawing lewis dot structures (lds) ionic ldss. Web for an anion, the lewis structure shows the gain of one or more electrons by the neutral atom, resulting in a negative charge. They also display the total number of lone pairs present in each of the atoms that constitute the molecule. Web draw the lewis dot diagram for a h−anion. Web lewis symbols for ionic compounds. Web draw the lewis dot diagram for a as anion. Web a lewis diagram shows how the valence electrons are distributed around the atoms in a molecule. Web this type of lewis dot structure is represented by an atomic symbol and a series of dots. From left to right as we face the page, there are 6, 6, and 2xx7 valence electrons associated with each atom, and this leads to our assignment of charge. Web a lewis diagram shows how the valence electrons are distributed around the atoms in a molecule. In almost all cases, chemical bonds are formed by interactions of valence electrons in. Neutral compounds ( 0) lewis dot structures: In section 4.7, we demonstrated that ions are formed by losing electrons to make cations, or by gaining electrons to form anions. To facilitate our understanding of how valence electrons interact, a simple way of representing those valence electrons would be useful. Web draw lewis structures for ionic compounds. Electronic structure is based. These symbols will represent the atoms present in the covalent bond. Many of the ions that form have eight electrons in their valence shell. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Web lewis symbols can be used to illustrate the formation of cations from atoms, as shown here for sodium and. Examples ionic compounds can be represented by the lewis dot structures, considering that a positive charge indicates that electrons have been lost. When drawing the structure of an ion, be sure to add/subtract electrons to account for. Web the following procedure will give you the correct lewis structure for any molecule or polyatomic ion that has one central atom. Web. Nitrate anion is an interesting customer in terms of resonance, in that three of the four atoms bear a formal electric charge in the resonance structure. Web draw the lewis dot diagram for a as anion. Web draw a lewis electron dot diagram for an atom or a monatomic ion. Web lewis structures (also known as lewis dot diagrams, electron. From left to right as we face the page, there are 6, 6, and 2xx7 valence electrons associated with each atom, and this leads to our assignment of charge. To facilitate our understanding of how valence electrons interact, a simple way of representing those valence electrons would be useful. Web for an anion, the lewis structure shows the gain of. Sigma & pi bonds ( 0) lewis dot structures: To facilitate our understanding of how valence electrons interact, a simple way of representing those valence electrons would be useful. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. In section 4.7, we demonstrated that ions are formed by losing electrons to make cations,. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Many of the ions that form have eight electrons in their valence shell. Since hydrogen is in group i it has one (1) valence electron in its shell. Exceptions ( 0) lewis dot structures: This problem has been solved! These symbols will represent the atoms present in the covalent bond. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Web a lewis diagram shows how the valence electrons are distributed around the atoms in a molecule. Ions ( 0) lewis dot structures: See the following examples for how to draw lewis dot. Web draw lewis structures for ionic compounds. To facilitate our understanding of how valence electrons interact, a simple way of representing those valence electrons would be useful. Web draw a lewis electron dot diagram for an atom or a monatomic ion. Web this type of lewis dot structure is represented by an atomic symbol and a series of dots. Likewise, they can be used to show the formation of anions from atoms, as shown here for chlorine and sulfur: Web lewis symbols for ionic compounds. In almost all cases, chemical bonds are formed by interactions of valence electrons in atoms. In the section above, we saw how ions are formed by losing electrons to make cations or by gaining electrons to form anions. Web a lewis diagram shows how the valence electrons are distributed around the atoms in a molecule. Web lewis structures (also known as lewis dot diagrams, electron dot diagrams,lewis dot formula lewis dot structures, and electron dot structures) are diagrams that show the bonding between atoms of a molecule and the lone pairs of electrons that may exist in the molecule. The astute reader may have noticed something: Web drawing lewis dot structures (lds) ionic ldss. Web for an anion, the lewis structure shows the gain of one or more electrons by the neutral atom, resulting in a negative charge. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Web draw the lewis dot diagram for a h−anion. From left to right as we face the page, there are 6, 6, and 2xx7 valence electrons associated with each atom, and this leads to our assignment of charge.
Lewis Dot Structures of Atoms and Ions YouTube
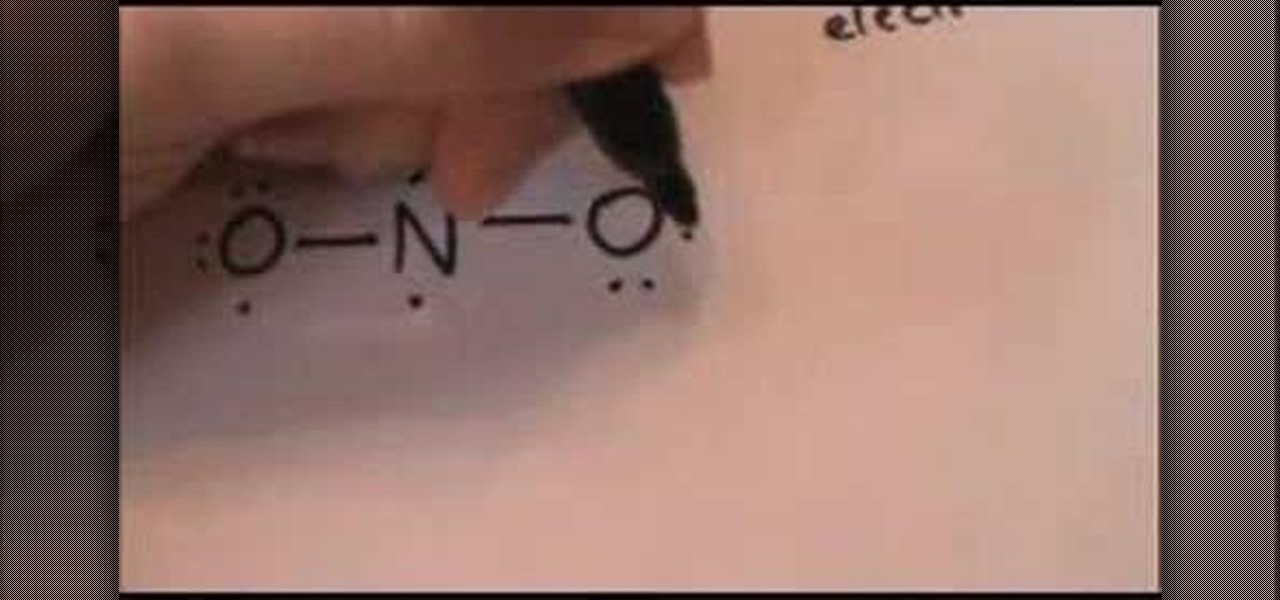
How to Draw a Lewis dot structure for nutrite anion « Science Experiments

How to Draw a Lewis Structure

Draw the Lewis structure for the polyatomic trisulfide anion. Be sure

Draw the Lewis Dot structure for the Phosphate anion?… SolvedLib

Comment dessiner une représentation de Lewis Wiki Chimie
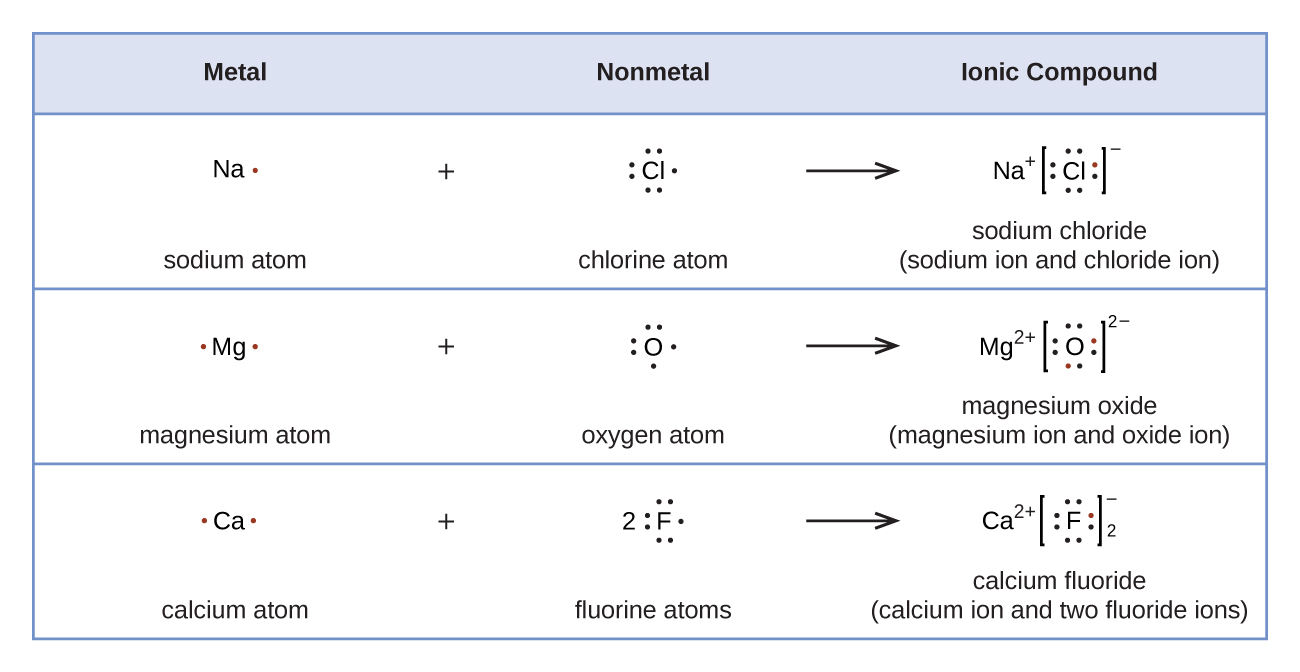
4.3 Lewis Symbols and Structures Chemistry LibreTexts
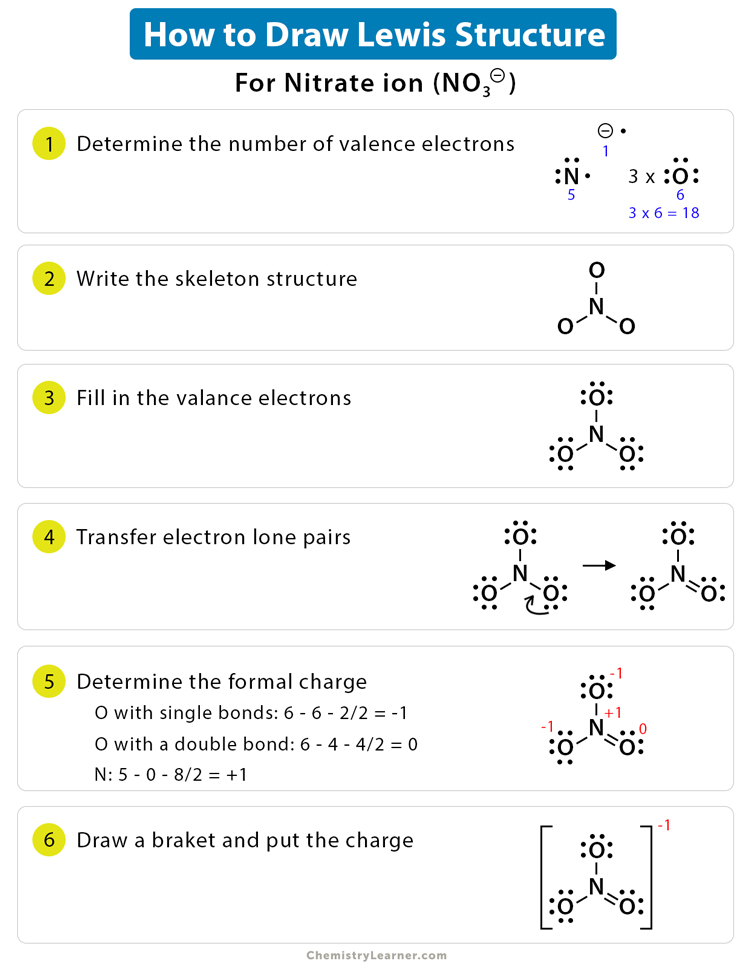
Lewis Dot Structure Definition, Examples, and Drawing
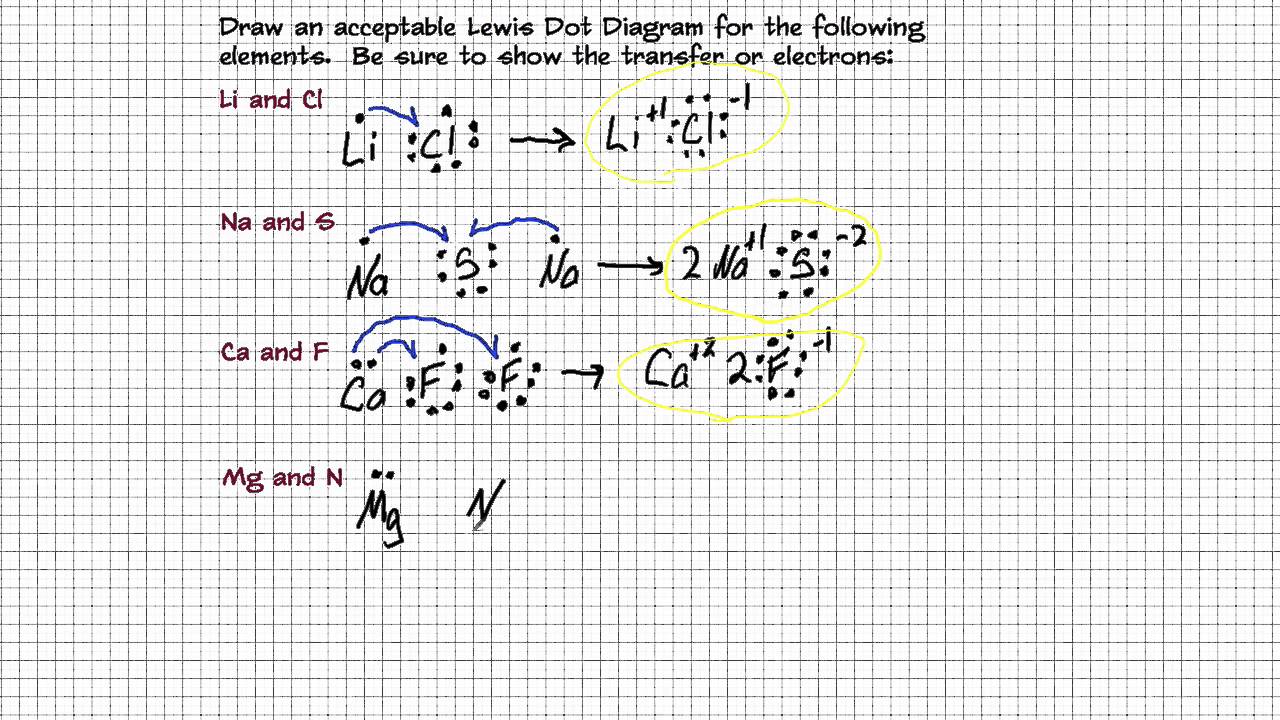
Drawing Lewis Dot Diagrams for Ionic Compounds YouTube

3 Ways to Draw Lewis Dot Structures wikiHow
Nitrate Anion Is An Interesting Customer In Terms Of Resonance, In That Three Of The Four Atoms Bear A Formal Electric Charge In The Resonance Structure.
Web Draw A Lewis Electron Dot Diagram For An Atom Or A Monatomic Ion.
Examples Ionic Compounds Can Be Represented By The Lewis Dot Structures, Considering That A Positive Charge Indicates That Electrons Have Been Lost.
Shared Pairs Of Electrons Are Drawn As Lines Between Atoms, While Lone Pairs Of Electrons Are Drawn As Dots Next To Atoms.
Related Post: