Draw The Lewis Structure For Hcn.
Draw The Lewis Structure For Hcn. - At least two lewis structures can be drawn for bcl 3. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Draw a skeleton structure put the least electronegative atom c in the middle with h and cl on either side. To draw the lewis structure of hcn, put carbon (c) in the middle, hydrogen (h) on one side, and nitrogen (n) on the other. In order to draw the lewis structure of hcn, first of all you have to find the total number of valence electrons present in the hcn molecule. For the hcn lewis structure, calculate the total number of valence electrons for the hcn. Put least electronegative atom in centre3. Put one electron pair in each bond4. How to draw a lewis structure for hcn? Figure out how many electrons the molecule must have, based on the number of valence electrons in each atom. Calculate the valence electrons the molecule of hcn consists of a hydrogen atom which has one valence electron in its outermost shell as it is a donor atom too, while next is a carbon atom that has four valence electrons participating. Fill out the octet of nitrogen with a lone pair. One should follow the given steps to draw the. Count the valence electrons you can use h + c + n =1 + 4 + 5 = 10 step 3. Here, the given molecule is hcn. Web =1+4+5 = 10 valence electrons. However, there are two possible resonance structures that can be drawn for hcn: Web the lewis structure of hcn shows a triple bond between the carbon and. Web drawing the lewis structure for hcn. Draw the lewis structure for hcn. 5) for the below compounds, draw the lewis structure, indicate the formal charge on each atom, determine the hybridization of all interior atoms, show the hybridization scheme, and sketch the orbital overlapping based on valance bond theory. Web the lewis structure (lewis dot diagram) for hcn.1. Web. Does this molecule exhibit resonance? Web this problem has been solved! 1.1k views 3 years ago chemistry. In this method, we find the bonds and lone pairs for the whole molecule, then plug it in to the atoms that we have to get the answer. Web =1+4+5 = 10 valence electrons. The sum of the valence electrons is. Include all lone pairs of electrons. At least two lewis structures can be drawn for bcl 3. Here, the given molecule is hcn. Put one electron pair in each bond4. This problem has been solved! Web to draw the lewis dot structure of any molecule, it is essential to know the total number of valence electrons in the structure. Draw a skeleton structure put the least electronegative atom c in the middle with h and cl on either side. Figure out how many electrons the molecule must have, based on. How to draw a lewis structure for hcn? To know the valence electrons of hcn, let us go through the valence electrons of individual atoms in hydrogen cyanide. The following procedure will give you the correct lewis structure for any molecule or polyatomic ion that has one central atom. Web hcn lewis structure: Put one electron pair in each bond4. Using arguments based on formal charges, explain why the. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. For the hcn lewis structure, calculate the total number of valence electrons for the hcn. Web 6 steps to draw the lewis structure of hcn step #1: However, there are two possible resonance structures that. Web 6 steps to draw the lewis structure of hcn step #1: Calculate the total number of valence electrons. Include all lone pairs of electrons. With the lewis structure for hcn you’ll need to share more than one pair of electrons between the carbon and the nitrogen atoms. You'll get a detailed solution from a subject matter expert that helps. Web drawing the lewis structure for hcn. 📌you can draw any lewis structures by following the simple steps mentioned.more Nov 12, 2017 here's how to do it. A) ccl4 b) n2h2 c) hcn 6) a) draw the molecular orbital diagram of the given compounds. Web the lewis structure of hcn shows a triple bond between the carbon and nitrogen atoms,. Here is a little flow chart of how we are going to do this: Web how to draw the lewis structure of hcn? How to draw a lewis structure for hcn? Draw at least one other lewis structure for the nitrate ion that is not plausible based on formal charges. Step method to draw the lewis structure of hcn. Does this molecule exhibit resonance? The second step is to add valence electrons to the one hydrogen atom, and the final step is to combine the. Include all lone pairs of electrons. Then, use single bonds to link carbon, hydrogen, and nitrogen. Web draw the most likely structure for hcn based on formal charges, showing the formal charge on each atom in your structure. A triple bond between the carbon and nitrogen atoms, with a lone pair of electrons on the nitrogen atom. Web steps to draw the lewis structure of hcn. Web the lewis structure (lewis dot diagram) for hcn.1. Now you can see that the central atom here is carbon because it is easy for carbon to become stable as it is the least electronegative of all. Add these electrons to give every atom an octet Does this molecule exhibit resonance?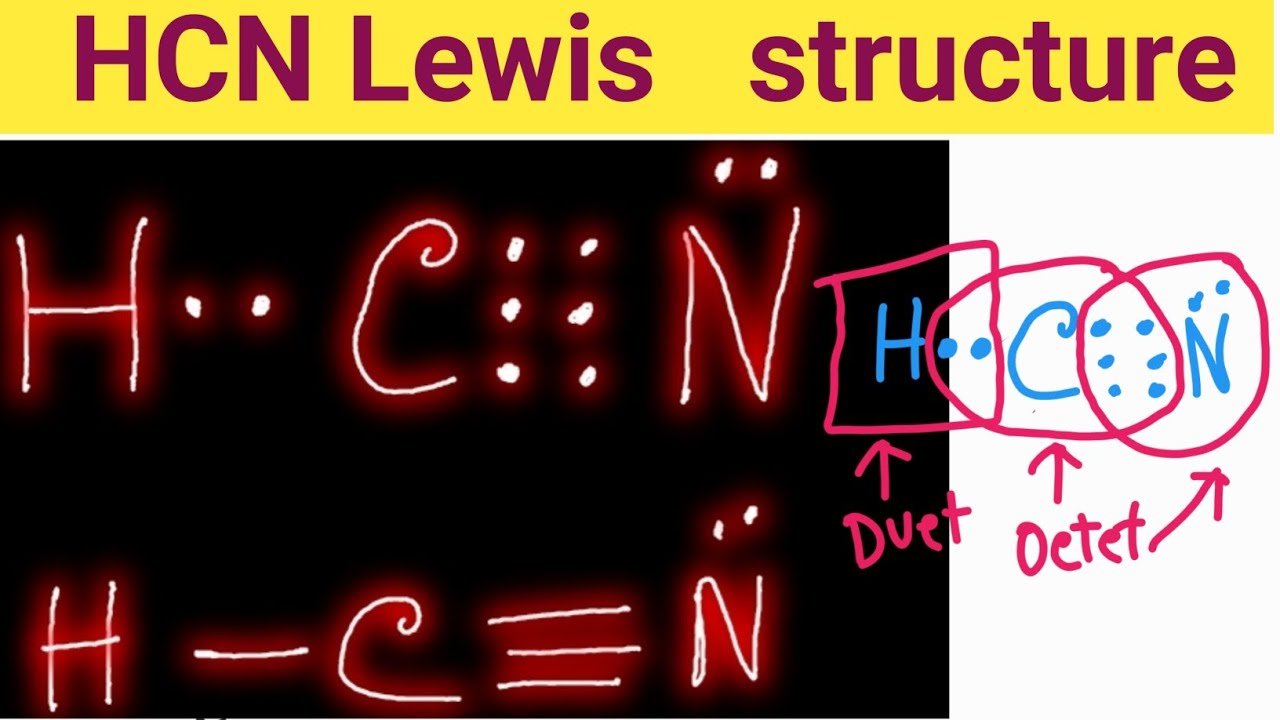
HCN Lewis StructureHydrogen Cyanide (HCN) Lewis Dot StructureDraw

Hcn Lewis Structure Bonds Draw Easy
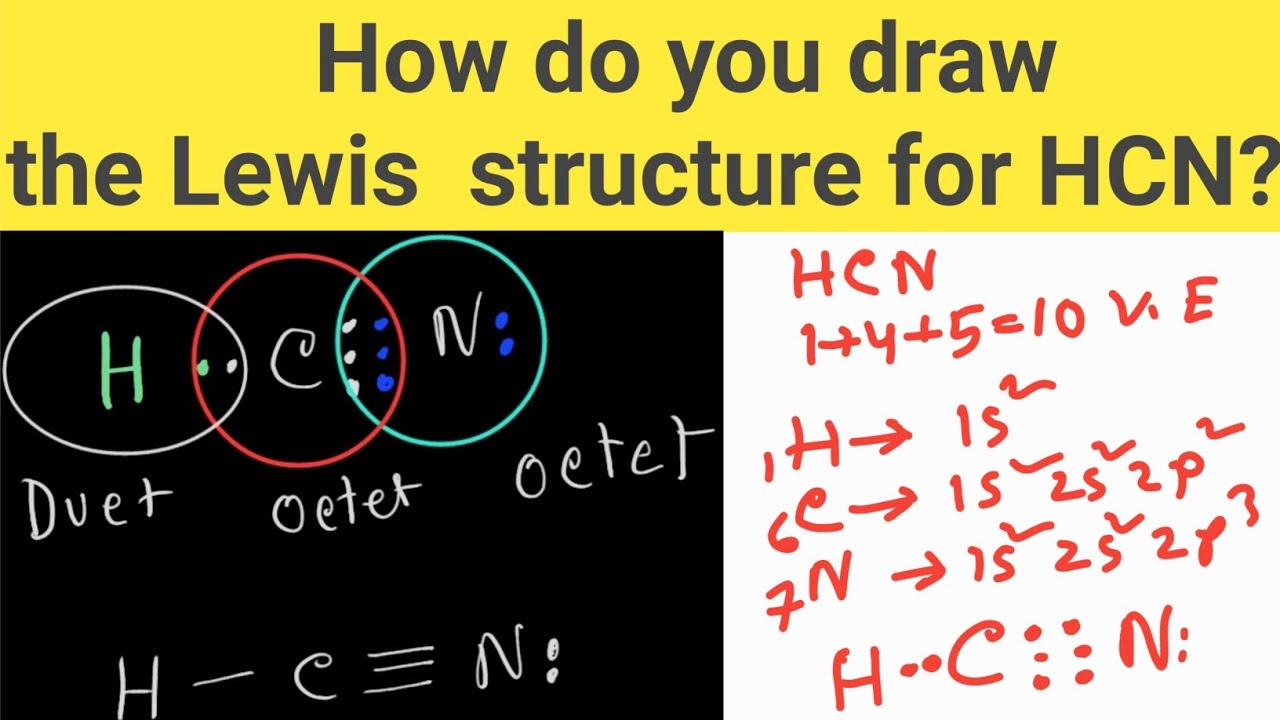
How do you draw the Lewis structure of HCN (hydrogen cyanide)? HCN

HCN Lewis Structure, Molecular Geometry, Hybridization, MO Diagram, and
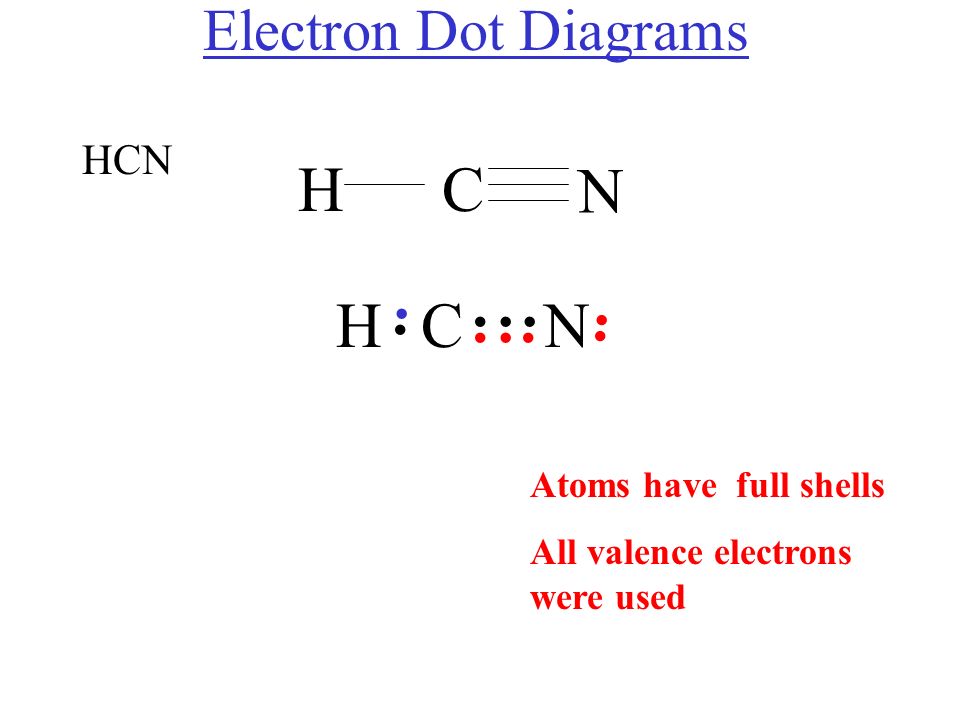
Lewis Diagram For Hcn

HCN Lewis Structure How to Draw the Dot Structure II lSCIENCE ll

HCN Lewis Structure (Hydrogen Cyanide) Molecules, Chemical formula, Lewis

Lewis Dot Diagram Of Hcn
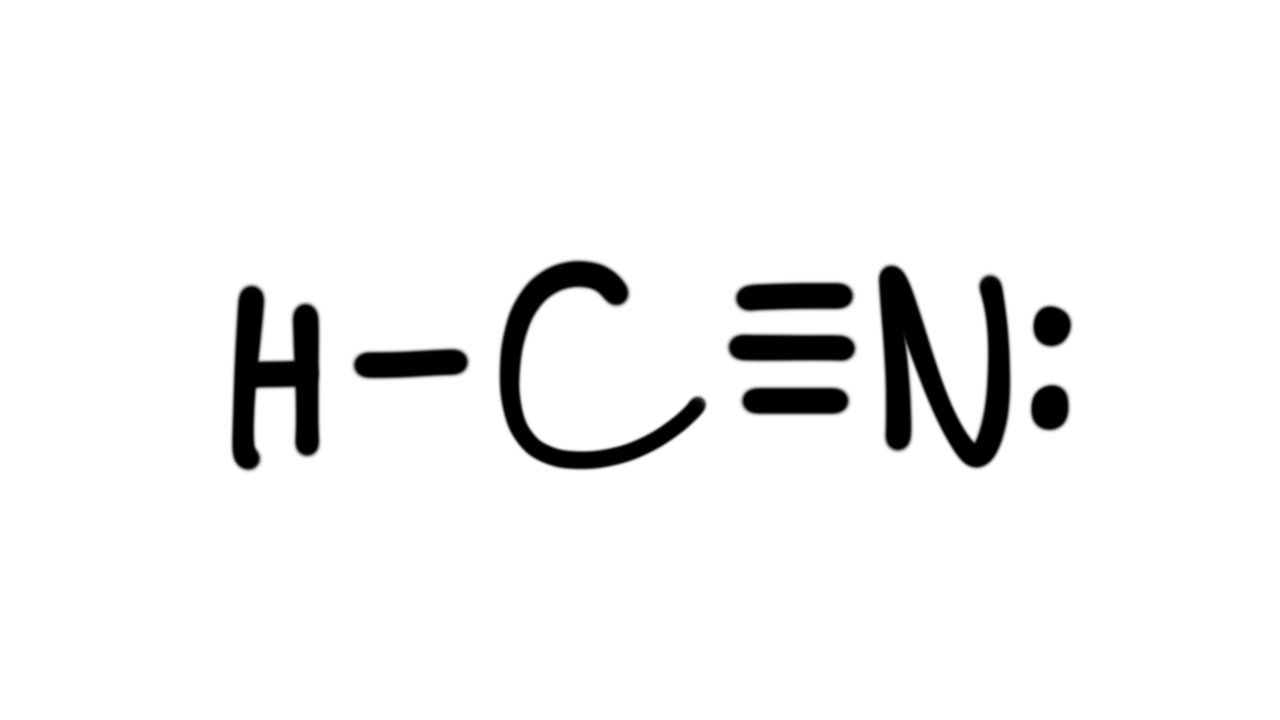
Lewis Diagram For Hcn
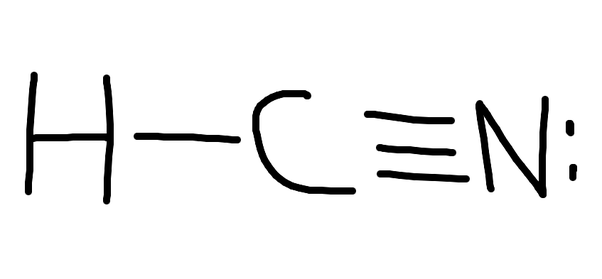
Lewis Diagram For Hcn
1.1K Views 3 Years Ago Chemistry.
You'll Get A Detailed Solution From A Subject Matter Expert That Helps You Learn Core Concepts.
Web Drawing Lewis Structures For Molecules With One Central Atom:
Here, The Given Molecule Is Hcn.
Related Post: