Draw The Lewis Structure For The Pcl+4 Ion
Draw The Lewis Structure For The Pcl+4 Ion - Shared pairs of electrons are drawn as lines between atoms, while lone pairs of electrons are drawn as dots next to atoms. Web draw lewis structures for covalent compounds. Show transcribed image text expert answer transcribed image text: The drive for eight the octet rule refers to the tendency of atoms to prefer to have eight electrons in the valence shell. Here are the steps and final. #1 draw a rough skeleton structure #2 mention lone pairs on the atoms #3 if needed, mention formal charges on the atoms now, let’s take a. For the pcl4+ structure use the periodic table to find the total number of valence electrons for the pcl4+ molecule. Web drawing lewis structures. Web identify the number of valence electrons in each atom in the nh 4 + ion. This problem has been solved! Web drawing lewis structures for molecules with one central atom: Web draw lewis structures for ionic compounds. Web draw lewis structures for covalent compounds. This problem has been solved! In the lewis structure of pcl 4+, there are four single bonds around the phosphorus atom, with four chlorine atoms attached to it, and on each chlorine atom, there are three. Web lewis structure of pcl 4+. For each, give (i) the molecular shape, (ii) the electron pair geometry at the central atom, and (iii) the hybridization of the central atom. Pcl 4+ has one phosphorus atom and four chlorine atoms. Web augchem 436 subscribers 1.8k views 3 years ago i needed to draw pcl4+ so i could show how to. Web drawing lewis structures for molecules with one central atom: Web augchem 436 subscribers 1.8k views 3 years ago i needed to draw pcl4+ so i could show how to input it into the chemdoodle chemical drawing program in owl. Web valence electronic structures can be visualized by drawing lewis symbols (for atoms and monatomic ions) and lewis structures (for. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Here are the steps and final. Web chemistry chemistry questions and answers draw the lewis structure for the (pcl4+)ion. Phosphorous and chlorine atoms have 5. Draw the lewis structure for the (pcl4+)ion. In the lewis structure of pcl 4+, there are four single bonds around the phosphorus atom, with four chlorine atoms attached to it, and on each chlorine atom, there are three lone pairs. Four pairs will be used in the chemical bonds between the p and f. This problem has been solved! When constructing a lewis diagram, keep in mind. A lewis diagram shows how the valence electrons are distributed around the atoms in a molecule. Web draw the lewis structures for each of the following ions or molecules. For very simple molecules and molecular ions, we can write the lewis structures by merely pairing up the unpaired electrons on the constituent atoms. Phosphorous and chlorine atoms have 5. In. When constructing a lewis diagram, keep in mind the octet rule, which refers to the tendency. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Pcl 4+ has one phosphorus atom and four chlorine atoms. Lewis structures are representations of molecules that include not only what atoms are present in the molecule but. Web pcl4+ lewis structure contains 32 total valence electrons. Also, there is a positive (+1) charge on the phosphorus atom. Web draw the lewis structure for the (pcl_(4)^(+))ion. Web draw the lewis structures for each of the following ions or molecules. Cl assign a formal charge to each atom in the student's lewis structure atom formal charge left cl top. Web draw the lewis structure for the pcl+4 ion. Web pcl4+ is a chemical formula for tetrachloro phosphanium ion. Web pcl4+ lewis structure contains 32 total valence electrons. For very simple molecules and molecular ions, we can write the lewis structures by merely pairing up the unpaired electrons on the constituent atoms. Lewis structures are representations of molecules that include. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Web chemistry chemistry questions and answers draw the lewis structure for the (pcl4+)ion. A lewis diagram shows how the valence electrons are distributed around the atoms in a molecule. Show transcribed image text expert answer transcribed image text: Web draw lewis structures for covalent. When constructing a lewis diagram, keep in mind the octet rule, which refers to the tendency. Web chemistry chemistry questions and answers draw the lewis structure for the (pcl4+)ion. Pcl 4+ has one phosphorus atom and four chlorine atoms. Phosphorous and chlorine atoms have 5. Shared pairs of electrons are drawn as lines between atoms, while lone pairs of electrons are drawn as dots next to atoms. Web draw the lewis structures for each of the following ions or molecules. #1 draw a rough skeleton structure #2 mention lone pairs on the atoms #3 if needed, mention formal charges on the atoms now, let’s take a. Web draw the lewis structure for the pcl+4 ion. Web augchem 436 subscribers 1.8k views 3 years ago i needed to draw pcl4+ so i could show how to input it into the chemdoodle chemical drawing program in owl. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Here are the steps and final. The following procedure can be used to draw lewis structure for simple molecules. Also, there is a positive (+1) charge on the phosphorus atom. Use the lewis electron structure of nh 4 + to identify the number of bonding and nonbonding electrons associated with each atom and then use equation 4.4.1 to calculate the formal charge on each atom. Lewis structures are representations of molecules that include not only what atoms are present in the molecule but also how the atoms are connected. In the lewis structure of pcl 4+, there are four single bonds around the phosphorus atom, with four chlorine atoms attached to it, and on each chlorine atom, there are three lone pairs.
How To Draw Lewis Structures Abilitystop

How to Draw a Lewis Structure

3 Ways to Draw Lewis Dot Structures wikiHow

How to Draw the Lewis Structure for PCl4+ YouTube
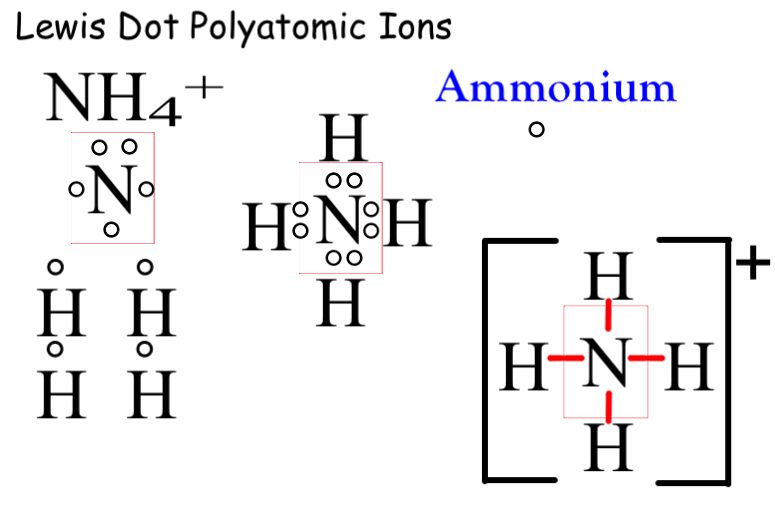
How do you draw lewis structures for polyatomic ions? Socratic

How To Draw Lewis Dot Structures For Compounds
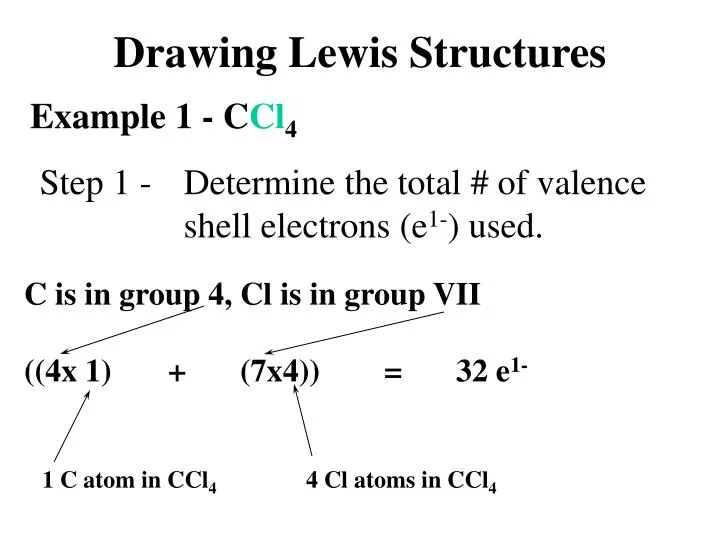
How To Draw Lewis Structures For Polyatomic Ions
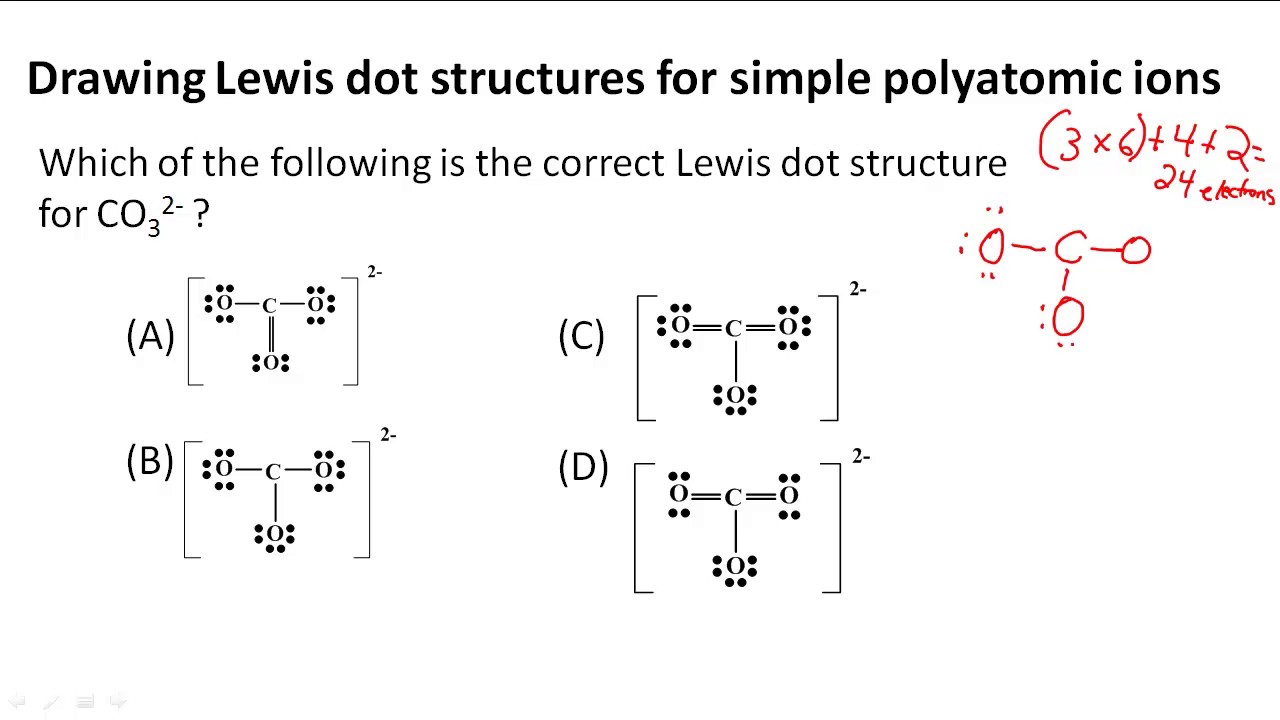
Lewis Structure Practice Worksheet
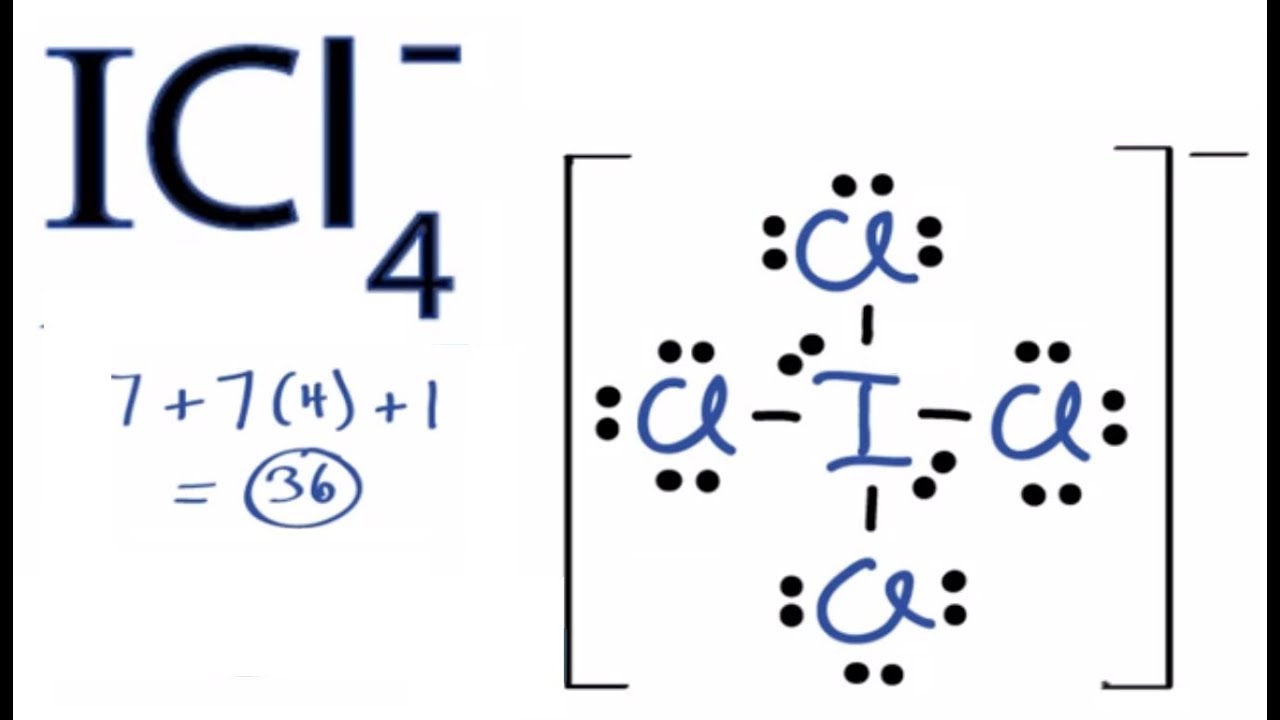
Draw the Lewis Structure for the Pcl+4 Ion
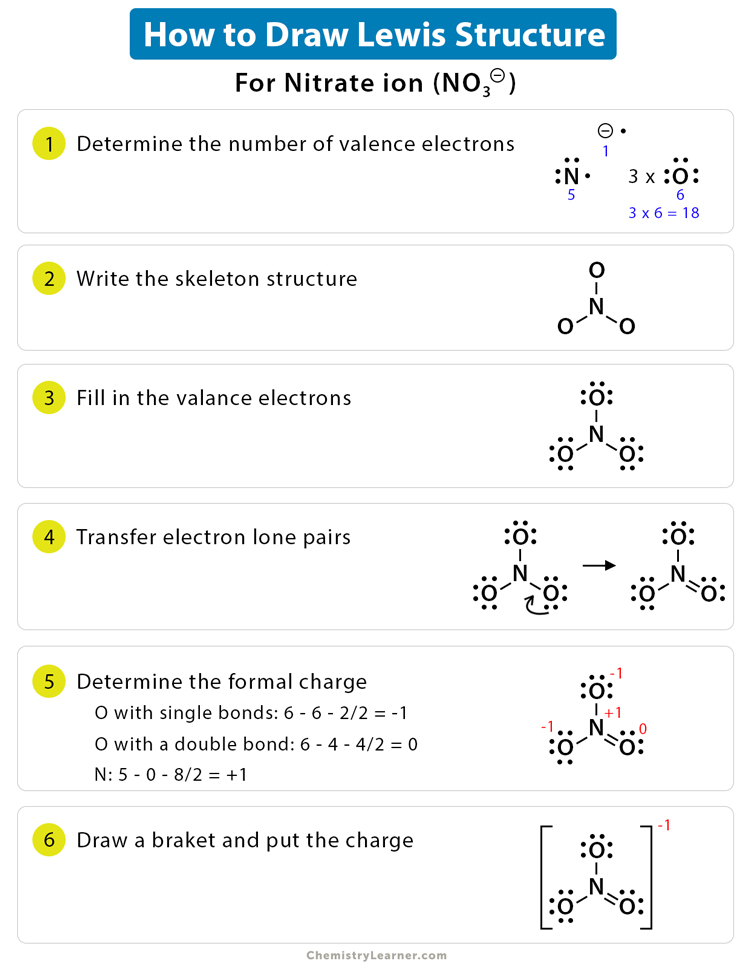
Lewis Dot Structure Definition, Examples, and Drawing
The Drive For Eight The Octet Rule Refers To The Tendency Of Atoms To Prefer To Have Eight Electrons In The Valence Shell.
A Lewis Diagram Shows How The Valence Electrons Are Distributed Around The Atoms In A Molecule.
You'll Get A Detailed Solution From A Subject Matter Expert That Helps You Learn Core Concepts.
For Very Simple Molecules And Molecular Ions, We Can Write The Lewis Structures By Merely Pairing Up The Unpaired Electrons On The Constituent Atoms.
Related Post: