Drawing D Orbitals
Drawing D Orbitals - Web an orbital is a space where a specific pair of electrons can be found. Web draw the shapes of various `p` and `d` orbitals. For an s orbital, draw a circle; Just as with the other orbitals, as number of nodal surfaces increases as you increase n. In two dimensions, we draw it as a circle. D orbitals are described only in terms of their energy, and f orbitals are only mentioned in passing. Web atomic orbitals are the wavefunctions which are solutions of the schroumldinger equation for the hydrogen atom the subset of atomic orbitals and are plotted in three dimensions to exhibit their characteristic shapes the orbitals are drawn by showing their boundary surfaces in the second view and signs are attached to the. The five d orbitals have m l values of −2, −1, 0, +1, and +2. S, p, d, and f. Web mo from d orbitals. Web the electrons in an atom occupy certain orbitals, which are divided into main types: The d orbitals are even higher in energy, and the f orbitals have the highest energy of all. This is also due to the history when they were discovered. Sigma ( σ σ ), pi ( π π ), and delta ( δ δ) bonds.. Web an orbital diagram, or orbital box diagram, is a way of representing the electron configuration of an atom. Web 1s from 2s, etc. Start from the third row, all the elements after sodium (na) have d orbitals. Web mo from d orbitals. In two dimensions, we draw it as a circle. For an f orbital, see below. You can puzzle them out from the rotating images, the dot density diagrams and the orbital surface diagrams if you like, but analysis of these orbitals is usually considered beyond the scope of general chemistry. Imagine shells around the nucleus, that get bigger and bigger. A box, line, or circle, is drawn to represent. Generally, there are three types of bonding and antibonding interactions that may occur with d d orbitals: 12k views 3 years ago. Web the electrons in an atom occupy certain orbitals, which are divided into main types: The orbital diagram or orbital notation simply represents the arrangement of electrons in different orbitals of an atom. Web atomic orbitals are the. The five d orbitals have m l values of −2, −1, 0, +1, and +2. Web an orbital is a space where a specific pair of electrons can be found. Web this page discusses atomic orbitals at an introductory level. Sigma ( σ σ ), pi ( π π ), and delta ( δ δ) bonds. One reason for this. Orbital diagrams must follow 3 rules: Web an orbital diagram, or orbital box diagram, is a way of representing the electron configuration of an atom. The d orbitals are even higher in energy, and the f orbitals have the highest energy of all. An s orbital is a sphere. Web beginning with the transition metal scandium (atomic number 21), additional. See resources for a diagram showing the filling order. S orbitalsthere is only 1 s orbital in each s subshell.shape of s orbital is spherica. The s orbitals are spherical and have the lowest energy, while the p orbitals are shaped like dumbbells and have slightly higher energy. One reason for this is that the f orbitals are very little. In an orbital diagram, an electron is represented by an arrow, while a box represents an atomic orbital. D orbitals are described only in terms of their energy, and f orbitals are only mentioned in passing. See resources for a diagram showing the filling order. Web an orbital diagram, or orbital box diagram, is a way of representing the electron. 1.7k views 2 years ago. Web 1s from 2s, etc. It explores s and p orbitals in some detail, including their shapes and energies. Just as with the other orbitals, as number of nodal surfaces increases as you increase n. Web an orbital is a space where a specific pair of electrons can be found. Imagine shells around the nucleus, that get bigger and bigger. Mdcat chemistry | mdcat chemistry tricks | how to prepare for. The s orbitals are spherical and have the lowest energy, while the p orbitals are shaped like dumbbells and have slightly higher energy. Web atomic orbitals are the wavefunctions which are solutions of the schroumldinger equation for the hydrogen. Just as with the other orbitals, as number of nodal surfaces increases as you increase n. Orbital diagrams must follow 3 rules: A box, line, or circle, is drawn to represent each orbital in the electron configuration (using the aufau principle to order the orbitals and hence the boxes, lines or circles, as shown below) 1s → 2s → 2p x 2p y 2p z → 3s → 3p x 3p y 3p z → The five equivalent 3d orbitals of the hydrogen atom. For a p orbital, draw a figure eight; See resources for a diagram showing the filling order. One reason for this is that the f orbitals are very little used in any chemical bonds. Web mo from d orbitals. There are three different rules used for constructing an atomic orbital diagram. The first principal shell to have a d subshell corresponds to n = 3. It explores s and p orbitals in some detail, including their shapes and energies. Start from the third row, all the elements after sodium (na) have d orbitals. S orbitalsthere is only 1 s orbital in each s subshell.shape of s orbital is spherica. D orbitals are described only in terms of their energy, and f orbitals are only mentioned in passing. Web four orbitals have six lobes oriented in various planes (easiest to draw). Web 1s from 2s, etc.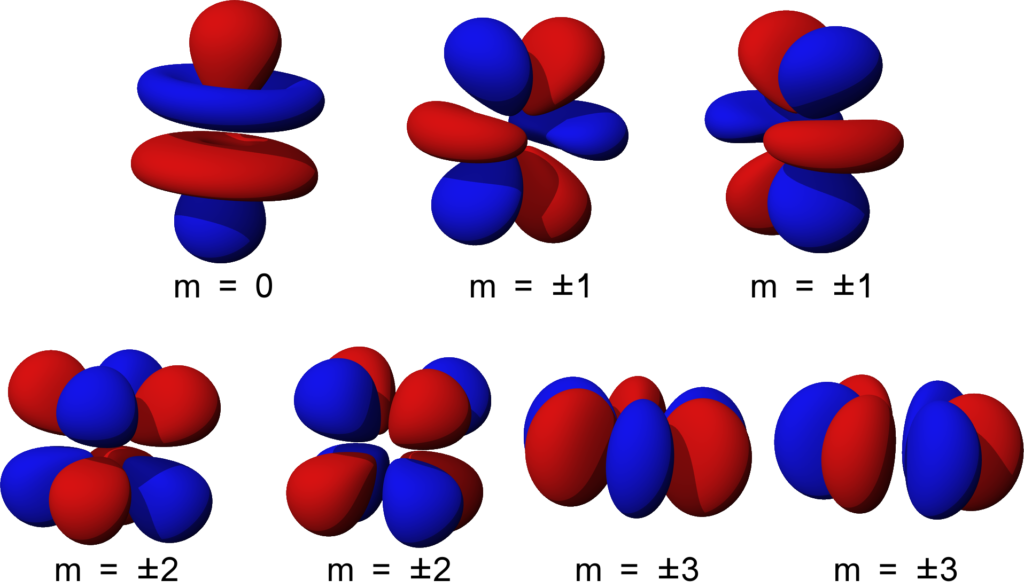
Shapes of Orbitals and their Types Chemistry Skills

how to draw shapes of d orbitals elliottlyde

1.The five different d atomic orbitals. The figure is adapted from
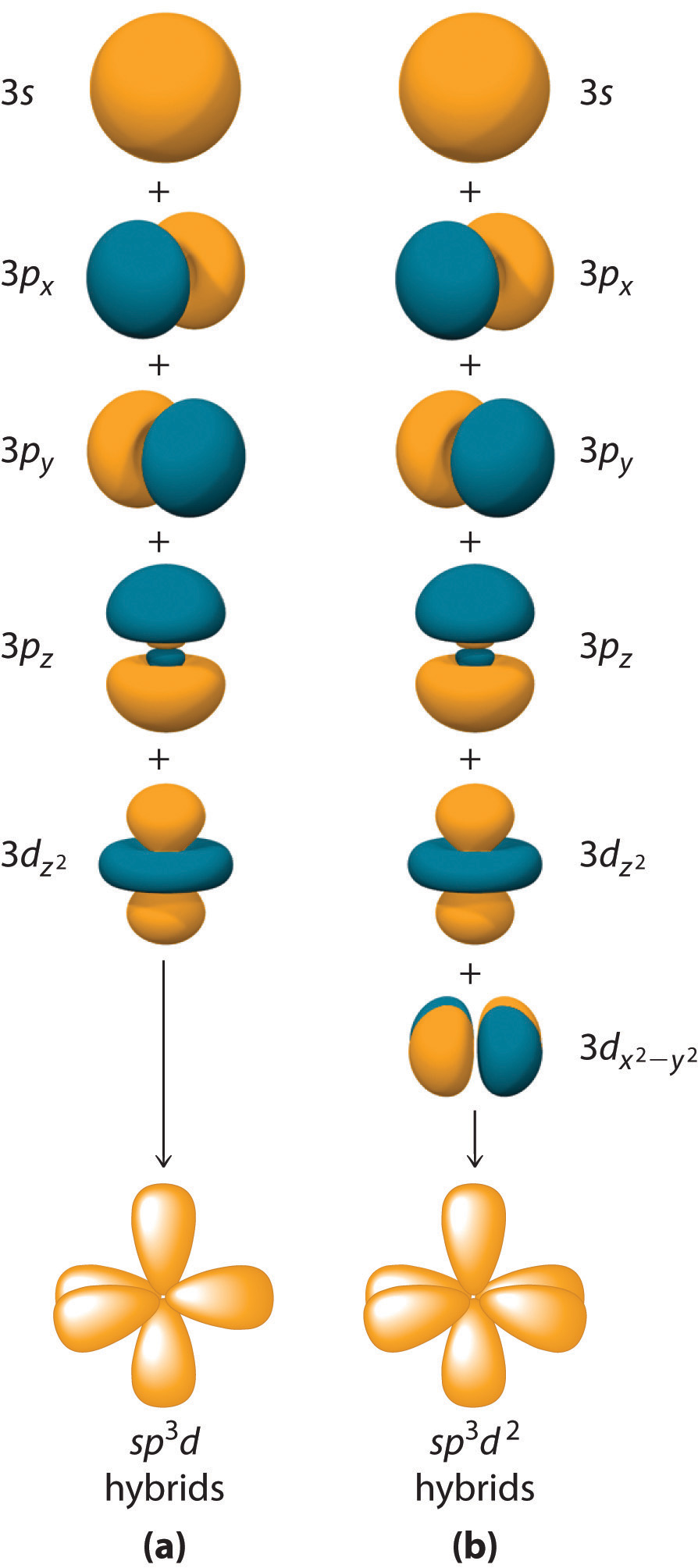
10.3 Hybridization of Atomic Orbitals Chemwiki

Shapes of Atomic Orbitals Shape of s, p, d, f Orbitals, FAQs, Examples

how to draw shapes of d orbitals elliottlyde
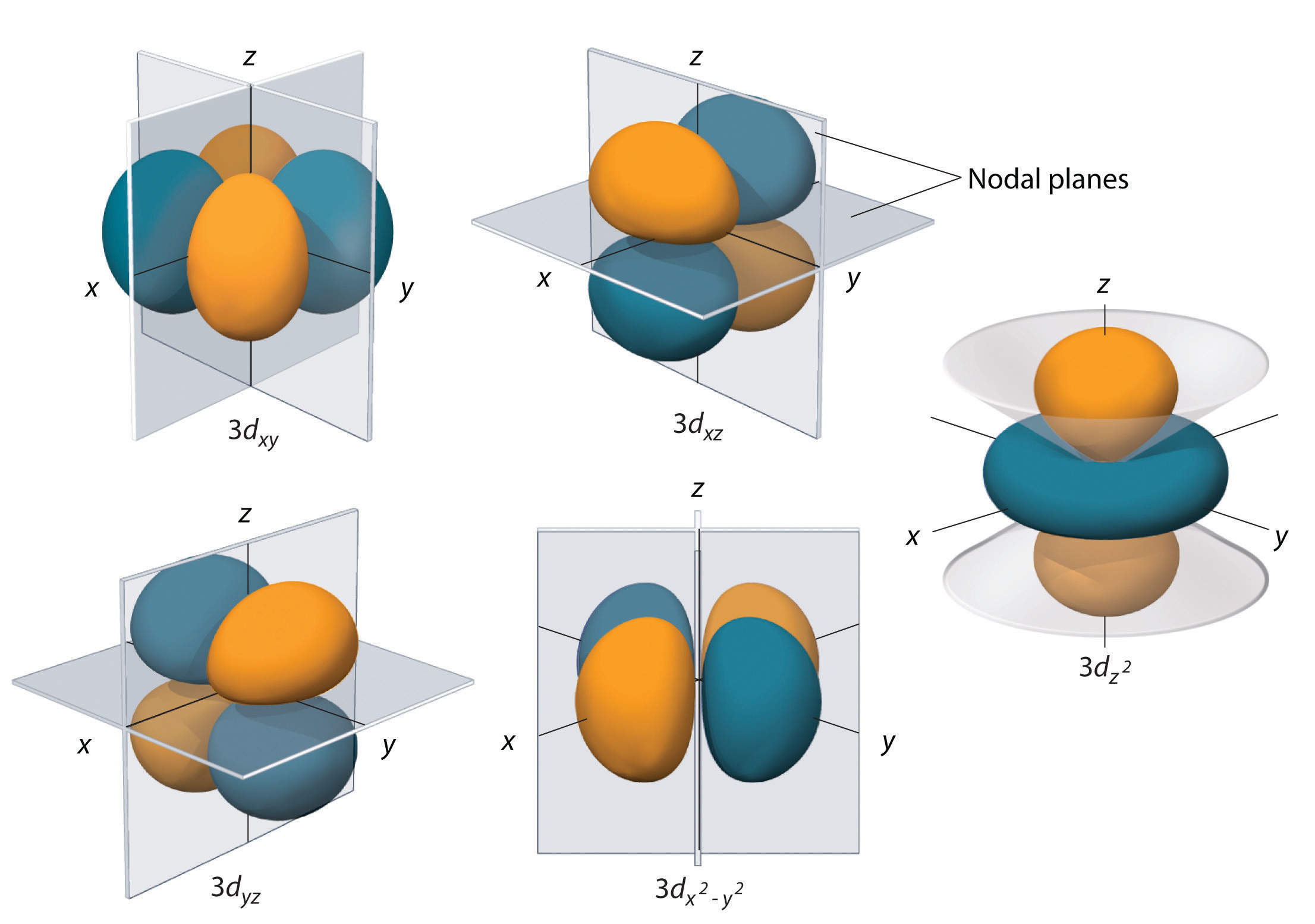
6.6 3D Representation of Orbitals Chemistry LibreTexts

Atomic Orbitals D
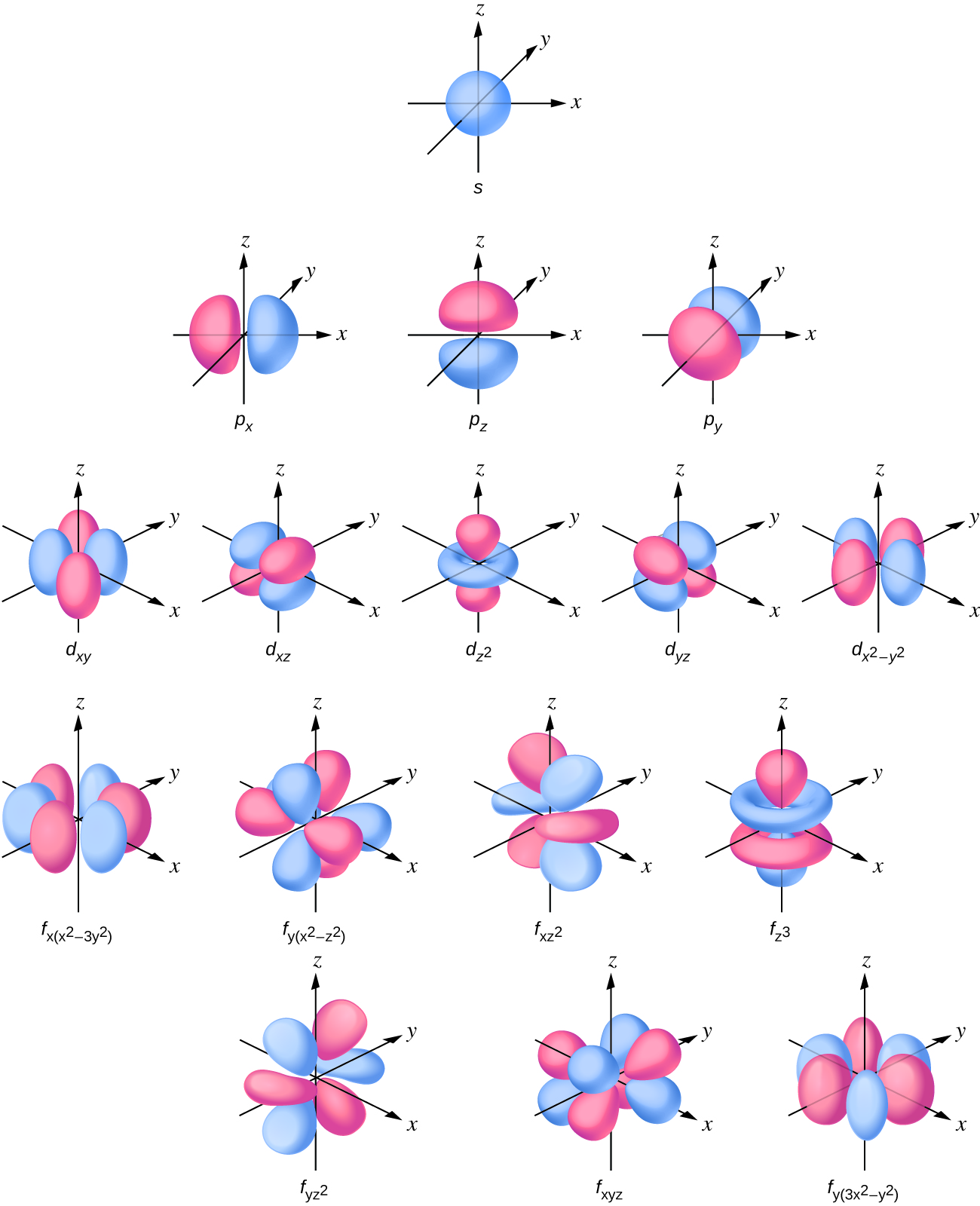
6.6 The Shapes of Atomic Orbitals Chemistry LibreTexts
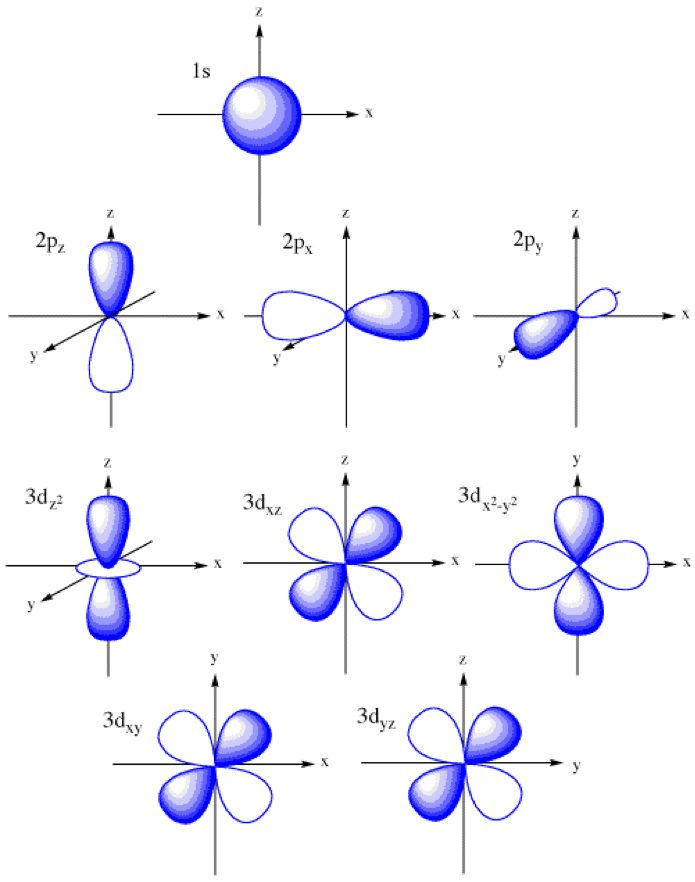
Atomic orbitals explained polizhuge
In An Orbital Diagram, An Electron Is Represented By An Arrow, While A Box Represents An Atomic Orbital.
S, P, D, And F.
Elements With D Orbitals, Especially The Transition.
The S Orbitals Are Spherical And Have The Lowest Energy, While The P Orbitals Are Shaped Like Dumbbells And Have Slightly Higher Energy.
Related Post: