Drawing Of Ionic Bond
Drawing Of Ionic Bond - A cation with a \(2+\) charge will make a stronger ionic bond than a cation with a \(1+\) charge. It starts by forming a bond between one or more than one atoms. At r0, the ions are more stable (have a lower potential energy) than they are at an infinite internuclear distance. Web it's just for ionic compounds electrons aren't shared so you won't have things like single bonds between atoms. This is because valence electrons are. Web how to draw the lewis structures of ionic compounds. Web i want to help you achieve the grades you (and i) know you are capable of; These grades are the stepping stone to your future. The astute reader may have noticed something: Web once they have mastered electron configuration diagrams, show your learners how they can adapt them to show structure and bonding in covalent and ionic compounds. A cation with a \(2+\) charge will make a stronger ionic bond than a cation with a \(1+\) charge. Many of the ions that form have eight electrons in their valence shell. Magnesium oxide dot & cross diagram Web an ionic compound is made up of charged particles, called ions. It has a giant lattice structure with strong electrostatic forces. Draw a square bracket around each ion. These grades are the stepping stone to your future. Web when drawing lewis dot structures for ionic compounds you need to follow a different set of rules than with lewis structures for covalent/molecular compounds. Even if you don't want to stud. When oppositely charged ions are brought together from r = ∞ to. Web how to draw the lewis structures of ionic compounds. Web we summarize the important points about ionic bonding: While you are learning how to draw dot and cross diagrams it’s useful to start with something you are already familiar with: We may have come across such questions while dealing with the chemistry subject. In section 4.7, we demonstrated that. Web draw lewis structures depicting the bonding in simple molecules thus far in this chapter, we have discussed the various types of bonds that form between atoms and/or ions. Look at the number of electrons on the outer shell of each atom. Web once they have mastered electron configuration diagrams, show your learners how they can adapt them to show. Web an ionic bond is the electrostatic force that holds ions together in an ionic compound. In ionic bonding, a simple process takes place. Drawing lewis structures for molecules with one central atom: A cation with a \(2+\) charge will make a stronger ionic bond than a cation with a \(1+\) charge. In section 4.7, we demonstrated that ions are. A cation with a \(2+\) charge will make a stronger ionic bond than a cation with a \(1+\) charge. Be sure to include the charges. These structural diagrams depict only the outer, or valence, shell electrons and are known as dot and cross diagrams. We may have come across such questions while dealing with the chemistry subject. Draw the ionic. The strength of ionic bonding depends on the magnitude of the charges and the sizes of the ions. Web an ionic bond is the electrostatic force that holds ions together in an ionic compound. Web we summarize the important points about ionic bonding: Draw the electron configuration diagrams for each atom in the compound. Drawing lewis structures for molecules with. The two most basic types of bonds are characterized as either ionic or covalent. Web follow your teacher’s directions to complete each ionic bond. Examples include nacl, mgf2, k2o,. Web when drawing lewis dot structures for ionic compounds you need to follow a different set of rules than with lewis structures for covalent/molecular compounds. Web draw lewis structures depicting the. Magnesium oxide dot & cross diagram Be sure to include the charges. Web i want to help you achieve the grades you (and i) know you are capable of; Web once they have mastered electron configuration diagrams, show your learners how they can adapt them to show structure and bonding in covalent and ionic compounds. The strength of the ionic. It is a fundamental topic that every chemistry enthusiast should know. Magnesium now has an empty third shell so draw the second shell instead. Web draw lewis structures depicting the bonding in simple molecules thus far in this chapter, we have discussed the various types of bonds that form between atoms and/or ions. Web representing a covalent bond using lewis. Web in ionic bonding, atoms transfer. Draw the ionic bonding in magnesium fluoride. Web draw lewis structures depicting the bonding in simple molecules thus far in this chapter, we have discussed the various types of bonds that form between atoms and/or ions. Web how to draw the lewis structures of ionic compounds. Web we summarize the important points about ionic bonding: How do we hold different ions together? Magnesium oxide dot & cross diagram The two most basic types of bonds are characterized as either ionic or covalent. For each ionic compound, you have been given the element names and the chemical formula. Draw the electron configuration diagrams for each atom in the compound. Web representing a covalent bond using lewis structures. Web when drawing lewis dot structures for ionic compounds you need to follow a different set of rules than with lewis structures for covalent/molecular compounds. These structural diagrams depict only the outer, or valence, shell electrons and are known as dot and cross diagrams. The strength of the ionic bond is directly dependent upon the quantity of the charges and inversely dependent on the distance between the charged particles. Look at the number of electrons on the outer shell of each atom. Electron configuration diagrams (see rsc.li/2whsi4f if you need a reminder).:max_bytes(150000):strip_icc()/ionic-bond-58fd4ea73df78ca1590682ad.jpg)
Examples of Ionic Bonds and Compounds

Ionic Bond Definition, Types, Properties & Examples
Examples of Ionic Bonds and Compounds
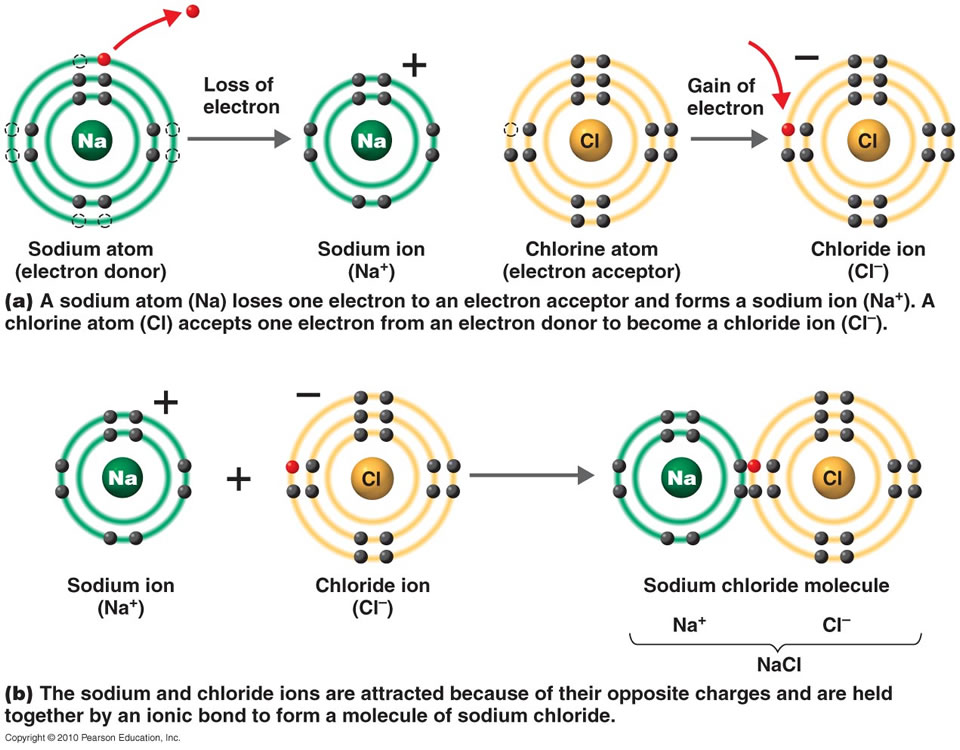
savvychemist Ionic Bonding (2) Dot and cross diagrams/Lewis structures

FileIonic bonding.svg Wikimedia Commons
Ionic Bond Vector Illustration Labeled Diagram With Formation

Ionic Bond Definition, Types, Properties & Examples
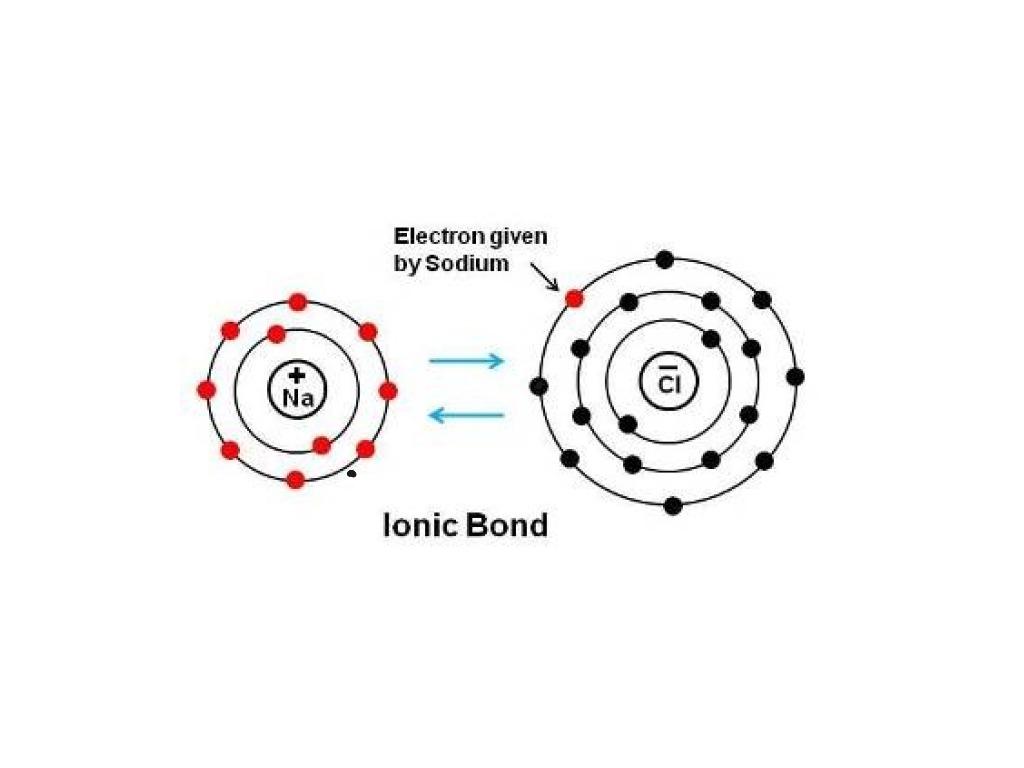
Ionic bond Science, Chemistry, Chemical Bonds ShowMe
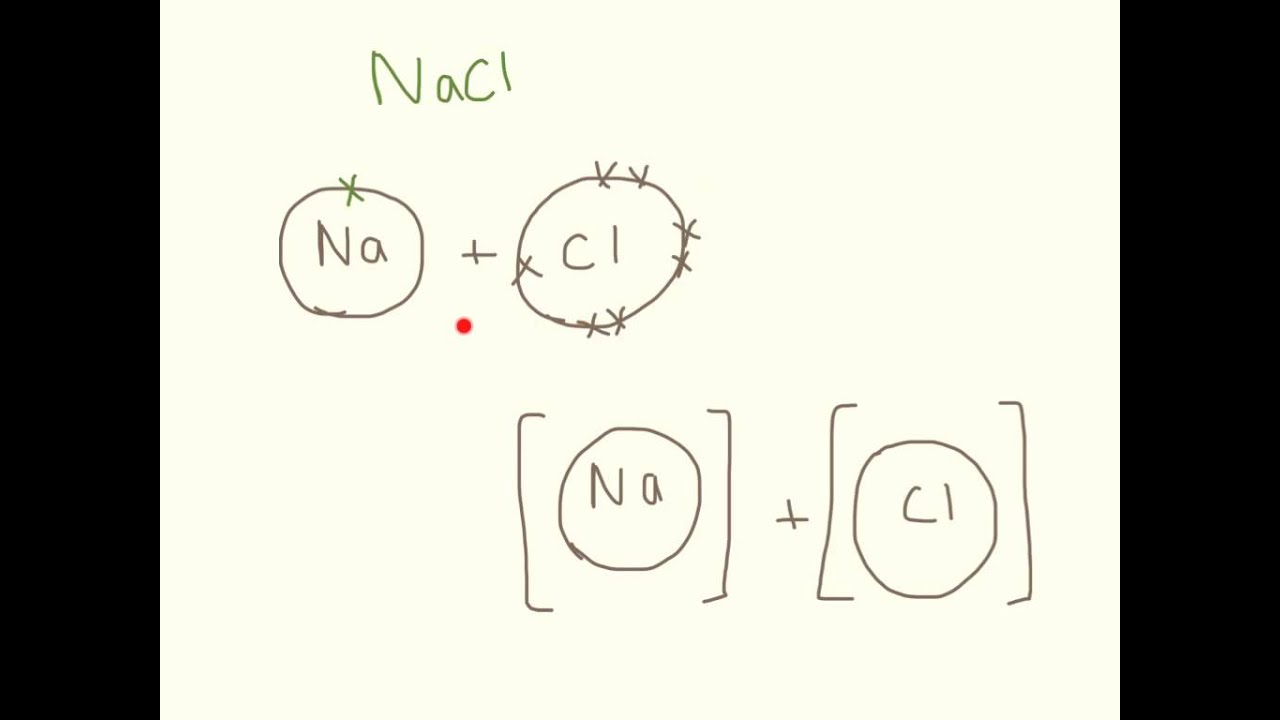
Drawing Ionic Bonding Dot and Cross Diagrams. YouTube
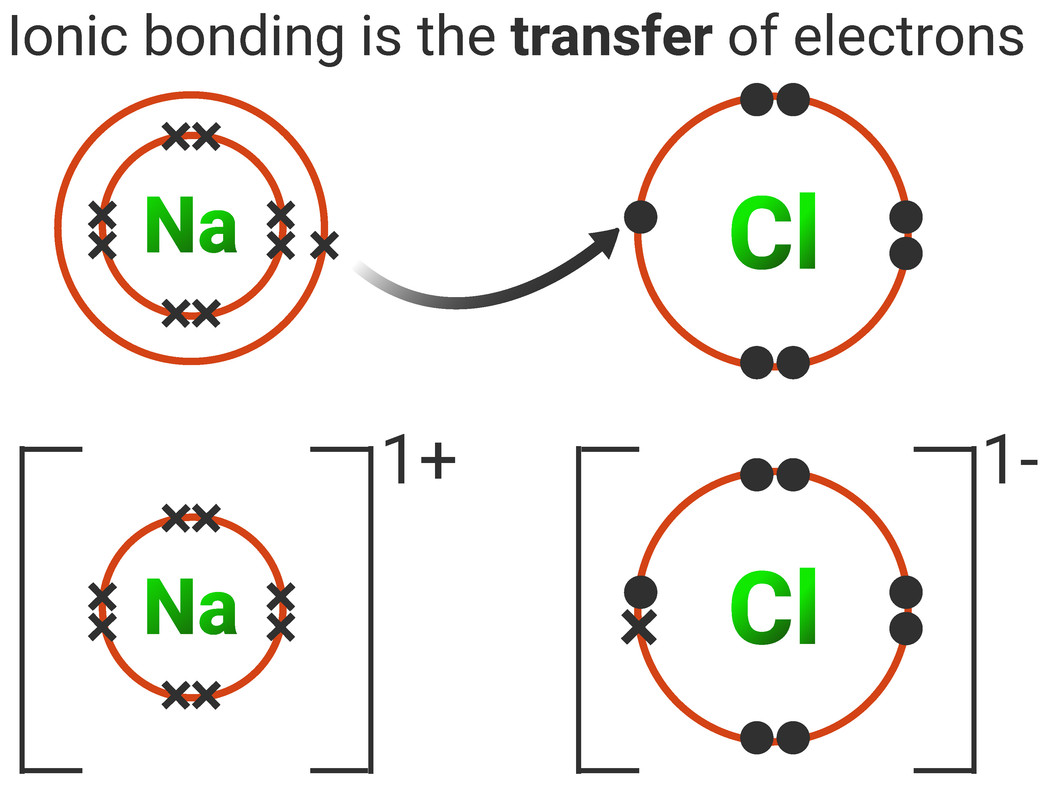
Bonding and Properties of materials OCR Gateway C2 revisechemistry.uk
The Strength Of Ionic Bonding Depends On The Magnitude Of The Charges And The Sizes Of The Ions.
1.21B Explain How To Draw Ionic Bonding.
It Has A Giant Lattice Structure With Strong Electrostatic Forces Of Attraction.
There Are Many Types Of Chemical Bonds And Forces That Bind Molecules Together.
Related Post:
