Specific Heat Drawing
Specific Heat Drawing - Heat energy = (mass of the object or substance) × (specific heat) × (change in temperature) q = m × c × \(\delta t\) or. The equation that relates heat (q) ( q) to specific heat (cp) ( c p), mass (m) ( m), and temperature change (δt) ( δ t) is shown below. Specific heats of common substances at 25 °c and 1 bar; Web the specific heat of a substance can be used to calculate the temperature change that a given substance will undergo when it is either heated or cooled. The amount of heat absorbed or released by a substance depends directly on the type of substance, its mass, and the. Web by following these steps and standards, you can create a mechanical drawing with specific heat treatment requirements, that conveys your design intent and specifications clearly and. The quantity of heat liberated or absorbed when the temperature of 1.00 gram of a substance falls or rises 1.00 c. The specific heat of water is 1 calorie (or 4.186 joules) per gram per celsius degree. The temperature change ( δt δ t) is the same in units of kelvins and degrees celsius (but not degrees fahrenheit). Its si unit is j/ (kg ⋅ ⋅ k) or j/ (kg ⋅ ⋅ °c °c ). C = \frac {q} {m \delta t} c = mδt q. Web specific heat and heat capacity are measures of the energy needed to change the temperature of a substance or object. The amount of heat absorbed or released by a substance depends directly on the type of substance, its mass, and the. Web specific heat online unit converter see. Web the symbol c stands for specific heat, and depends on the material and phase. H 2 o(s) 2.093 (at −10 °c) water vapor: The temperature change ( δt δ t) is the same in units of kelvins and degrees celsius (but not degrees fahrenheit). There are reasons he teaches this way and they are all good. The specific heat. The amount of heat absorbed or released by a substance depends directly on the type of substance, its mass, and the. C 2 h 6 o(l) 2.376: Q = cp × m × δt q = c p × m × δ t. Metals and semimetals common liquids and fluids c) = 5/9 [t ( c)] (9/5) + 32 the. Web the si unit for specific heat is j / (kg × k) or j / (kg ×oc). Web specific heat and heat capacity are measures of the energy needed to change the temperature of a substance or object. Web sal was explaining the intuition behind the formula for thermal conductivity, he was not solving for specific heat ratios. Where. Namely, by measuring the heat capacity of a sample of the substance, usually with a calorimeter, and dividing by the sample's mass. Web sal was explaining the intuition behind the formula for thermal conductivity, he was not solving for specific heat ratios. 1.00 cal = 4.184 j specific heat (c s): Web this chemical property, known as specific heat, is. The specific heat of water is 1 calorie (or 4.186 joules) per gram per celsius degree. Therefore, it requires 903 j to raise 1.00 kg of aluminum by 1.00 k. Web by following these steps and standards, you can create a mechanical drawing with specific heat treatment requirements, that conveys your design intent and specifications clearly and. The specific heat. Heat capacity and specific heat C 2 h 6 o(l) 2.376: Web this chemical property, known as specific heat, is defined as the amount of thermal energy needed to raise the temperature of 1 gram of a substance by 1 degree celsius. Namely, by measuring the heat capacity of a sample of the substance, usually with a calorimeter, and dividing. 1.00 cal = 4.184 j specific heat (c s): Web this chemical property, known as specific heat, is defined as the amount of thermal energy needed to raise the temperature of 1 gram of a substance by 1 degree celsius. Therefore, it requires 903 j to raise 1.00 kg of aluminum by 1.00 k. Unit of heat most commonly used. The choice is yours 路 ♂️ most of us have either been there or still stuck there. C = \frac {q} {m \delta t} c = mδt q. Specific heat is temperature (and phase) dependent. Web the specific heat is the amount of heat necessary to change the temperature of 1.00 kg of mass by 1.00 ºc. Heat energy =. Web specific heat online unit converter. Web the symbol c stands for specific heat and depends on the material and phase. Heat capacity and specific heat H 2 o(s) 2.093 (at −10 °c) water vapor: (recall that the temperature change δt is the same in units of kelvin and degrees celsius.) values of specific heat must generally be measured, because. Q = cp × m × δt q = c p × m × δ t. C 2 h 6 o(l) 2.376: Substance symbol (state) specific heat (j/g °c) helium: Web the specific heat of a substance can be used to calculate the temperature change that a given substance will undergo when it is either heated or cooled. Where q is the heat gained or lost, m is the mass of the object, c is its specific heat, and δt is the change in. The choice is yours 路 ♂️ most of us have either been there or still stuck there. Web the formula for specific heat looks like this: Web this chemical property, known as specific heat, is defined as the amount of thermal energy needed to raise the temperature of 1 gram of a substance by 1 degree celsius. The specific heat c is a property of the. Web specific heat 8/4/2018 2revised joule (j): The units of specific heat are usually calories or joules per gram per celsius degree. Therefore, it requires 903 j to raise 1.00 kg of aluminum by 1.00 k. There are reasons he teaches this way and they are all good. The specific heat is the amount of heat necessary to change the temperature of 1.00 kg of mass by 1.00ºc. C = \frac {q} {m \delta t} c = mδt q. Heat energy = (mass of the object or substance) × (specific heat) × (change in temperature) q = m × c × \(\delta t\) or.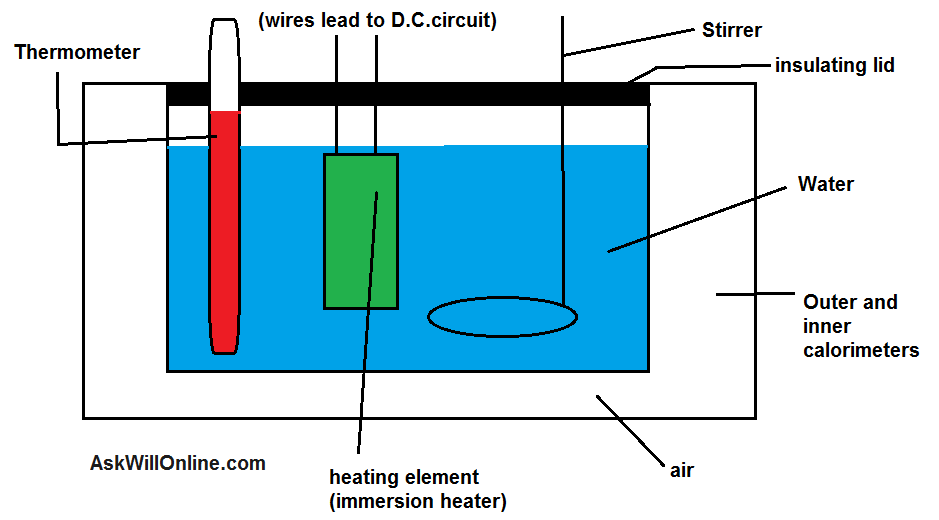
Specific Heat Capacity and Latent Heat Experiments In Physics Ask
To Determine Specific Heat Capacity Of A Given Solid Physics Practical

How To Calculate Specific Heat Of Metal Haiper

1.3 Specific Heat Capacity
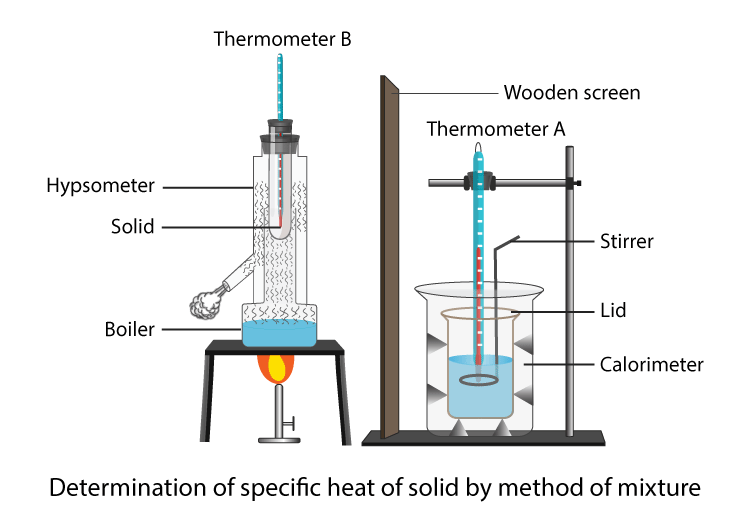
Finding a material's specific heat capacity GCSE Science Marked by

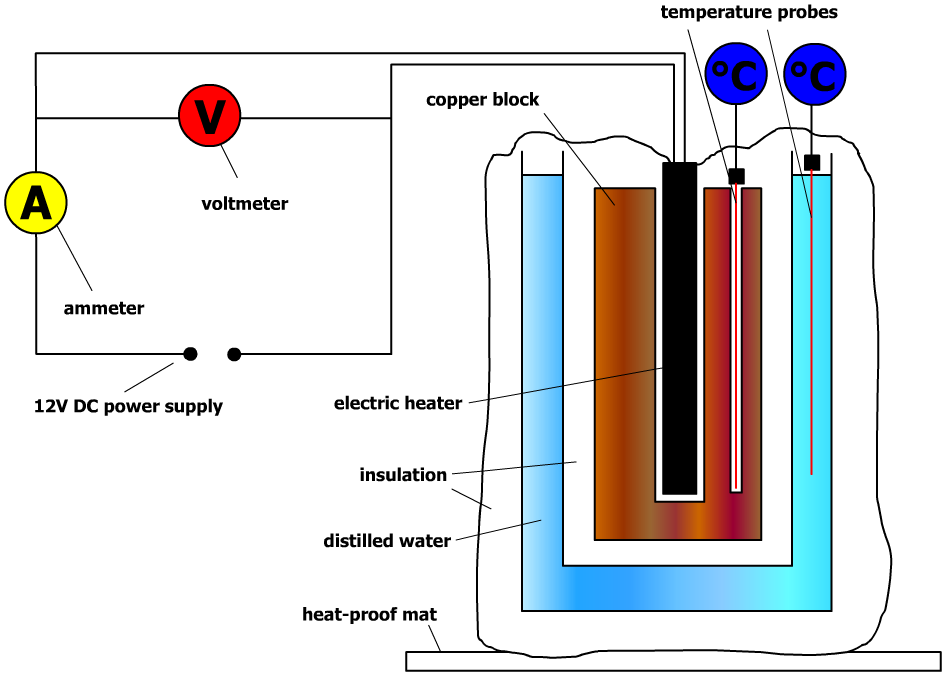
AQA A Level Physics Year 2 /IB Physics Specific Heat Capacity
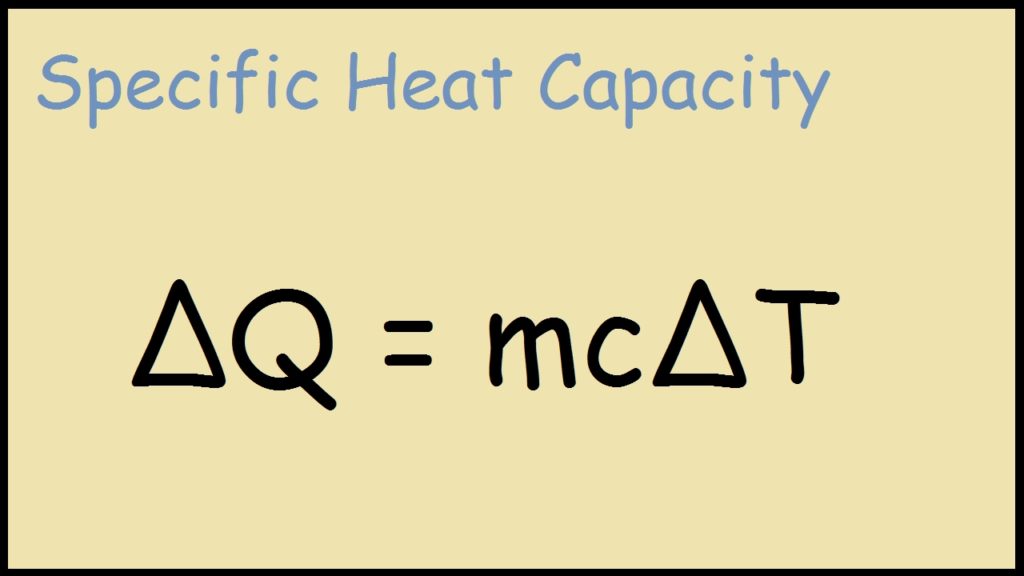
Specific Heat Formula Definition, Equations, Examples
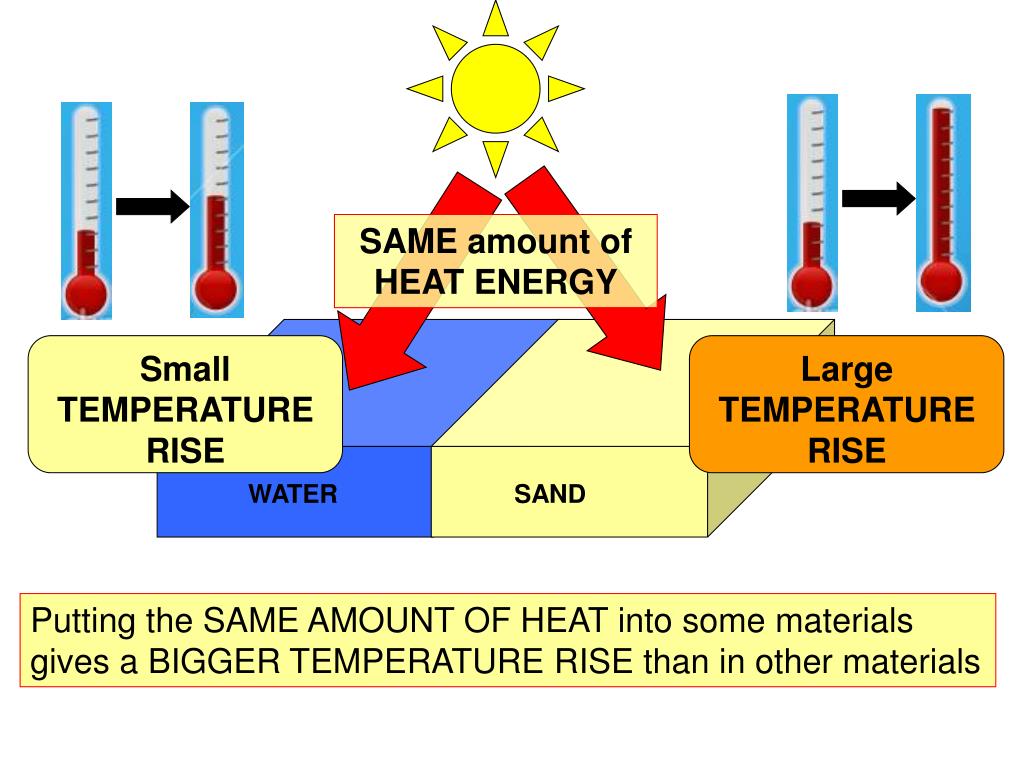
PPT SPECIFIC HEAT CAPACITY PowerPoint Presentation, free download

Lesson 10 Specific Heat YouTube
Lesson Video Specific Heat Capacity Nagwa
(Recall That The Temperature Change Δt Is The Same In Units Of Kelvin And Degrees Celsius.) Values Of Specific Heat Must Generally Be Measured, Because There.
Web Specific Heat Online Unit Converter See Also Tabulated Values For Gases, Food And Foodstuff, Metals And Semimetals, Common Liquids And Fluids And Common Solids, As Well As Values Of Molar Specific Heat For Common Organic Substances And Inorganic Substances.
1.00 Cal = 4.184 J Specific Heat (C S):
Web Sal Was Explaining The Intuition Behind The Formula For Thermal Conductivity, He Was Not Solving For Specific Heat Ratios.
Related Post: